In collaboration with Metabolix
Viscotek has been a strong advocate of good chromatography as a prerequisite for GPC (Gel Permeation Chromatography) data accuracy. Our recent work in application development has been driven by extremely difficult samples from industrial, biopharmaceutical and academic sources. These samples present challenges ranging from sample solubility, column adsorption as well as detection issues. This report will attempt to highlight a new approach that will be very helpful in certain advanced GPC applications where the eluent is expensive or hazardous.
SELS emphasizes the separate and different roles of sample solvent and mobile phase in a GPC analysis where the two liquid mediums can be synergistically applied to solve a variety of chromatographic problems. (Figure 1) This is a break from tradition where most GPC analyses involve a single solvent for sample prep and as the mobile phase. In SELS, one can choose the best solvent for a set of samples in term of solubility. On the other hand, users can also maximize detector sensitivity, particularly LS, by choosing a mobile phase that will provide the highest dn/dc values for the samples. Because of the much larger volumes required for the mobile phase, material cost and HS&E properties could also be important factors. Finally, column-sample interaction can also be moderated through proper selection of the mobile phase. In general, SELS is rapidly becoming a significant parameter in GPC method development.
The following is an excellent SELS example where we have applied this technique to a Polyhydroxy Alkanoate (PHA, Trade Name Mirel) material. Mirel is a family of biobased, sustainable and biodegradable plastics that are environmentally responsible alternatives to a wide variety of petroleum-based plastics. Mirel production reduces the use of fossil energy by more than 95% and provides a 200% reduction in greenhouse gases compared to production of conventional petroleum-based plastics. Mirel bioplastics can also harmlessly biodegrade at the end of their useful life in a broad range of environments including: soil, home compost, industrial compost, wetlands and the ocean. Metabolix is currently

|
producing injection molding, sheet and blown film grades, and focusing on short term applications such as: compost bags, horticulture and agriculture, business products, packaging, gift cards and electronic components.
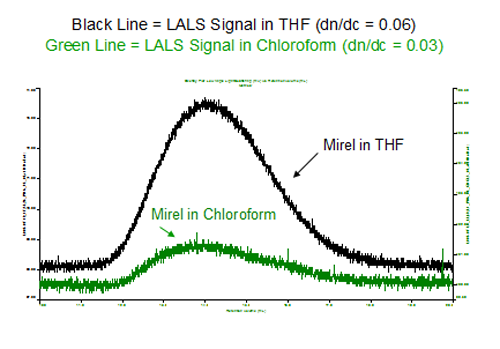
Mirel presented an interesting challenge to the Light Scattering GPC measurement. While Mirel is chloroform soluble, the dn/dc of this material is very low in chloroform. It is important to increase the dn/dc value in order to get better LS response. This can be achieved with a lower refractive index mobile phase like tetrahydrofuran (THF) as shown in figure 2. The PHA polymer is not soluble in THF, however does not come out of solution with THF as eluent during the timescale of the elution. Using the SELS concept, we combine of the solubility capability of chloroform and the enhanced LS response offered by THF in one single experiment to maximize the accuracy and the reliability of the analysis. The overlay of the Low Angle Laser Light Scattering (LALS) responses of Mirel in THF and chloroform clearly shows the power of SELS (figure 2). Typical dn/dc values in these two solvents are also given.
|
Authors:
Wei Sen Wong, PhD, Viscotek Corporation
Chris Mirley, PhD, Metabolix
Sean Daughtry, MS, Metabolix