Dynamic light scattering has been vastly explored as a tool for micelles characterization, not only for their size and stability, but also to determine the critical micellization concentration (CMC) and temperature (CMT)1-3. As a result of the unimers aggregation, as a function of the temperature for the case of this study, an increase in the scattered intensity is expected due to larger particles scattering substantially more photons. It’s been reported that for light-scattering techniques to be used reliably as CMC or CMT determination techniques, long correlation times are needed 1,4,5 which make this time-consuming and hence it’s not usually considered as a primary technique. We suggest in this application note, that DLS can be used in conjugation with other techniques to determine the CMT in a highly reproducible and time-efficient manner.
Dynamic light scattering has been vastly explored as a tool for micelles characterization, not only for their size and stability but also to determine the critical micellization concentration (CMC) and temperature (CMT)1-3. As a result of the unimers aggregation, as a function of the temperature for the case of this study, an increase in the scattered intensity is expected due to larger particles scattering substantially more photons. It’s been reported that for light-scattering techniques to be used reliably as CMC or CMT determination techniques, long correlation times are needed 1,4,5 which makes this time-consuming and hence it’s not usually considered as a primary technique. We suggest in this application note, that DLS can be used in conjugation with other techniques to determine the CMT in a highly reproducible and time-efficient manner. For a more comprehensive introduction on the topic and how the Zetasizer Nano could be used for CMC determination, the readers are referred to Ref. [1] and to Malvern Panalytical’s previous application note “Surfactant micelle characterization using dynamic light scattering”.
All measurements described in this application note were performed with Malvern Panalytical’s Zetasizer Ultra. The surfactant used was Pluronics F-68 (poloxamer 188, Sigma), a triblock co-polymer composed of polyethylene oxide (PEO) and polypropylene oxide (PPO) with a general structure of PEO-PPO-PEO. This polymer was selected due to it showing a transition from unimer to micelle at relatively high temperatures, and therefore its CMT could be investigated by DLS. For the CMT determination, the derived count rate was monitored at a back-scattering angle of detection, at 1°C increments from 40°C – 70°C. Adaptive Correlation was used to reduce the effect of transient events (contaminants, dust particles and aggregates) on the final result and allow for a more sensitive determination of the unimer/micelle transition point. This was performed for 5 different concentrations, ranging from 1.25 mg/mL to 20 mg/mL, to evaluate the effect of concentration on the CMT. A multi-angle dynamic light scattering measurement (back, side and forward scatter detectors) was also run to try to resolve the transition stage, which requires a higher resolution size analysis, and these were reported as intensity-weighted distribution plots. Each temperature point was performed in triplicates.
A 10 mg/mL solution of F-68 in water was first analyzed on the Zetasizer Ultra at different temperature points. Considering F-68 known behavior of forming micelles at higher temperatures it was expected that the derived count rate (the count rate obtained taking into consideration the attenuation factor used), would show an inflection point at the temperature where micelles start to form. This would be accentuated due to larger particles scattering considerably more photons, since according to Rayleigh theory intensity = diameter6, for Rayleigh scatterers (dh < 1/10 λlaser).
The effect in the derived count was substantial and the inflection point (or the CMT) was easily discernible at the intersect between the two lines, as demonstrated in Figure 1. For the initial temperature range, from 40°C – 51°C the count rate did not vary significantly, and hence a straight line was applied to the data points. Conversely, from 52°C upwards, a rapid increase in the number of photons detected occurred, and to this part of the data points, a 3rd degree polynomial was applied, since it showed the highest correlation coefficient value (R2 = 0.9989). The CMT can then be identified as the temperature at which the two lines intersect, 52°C.
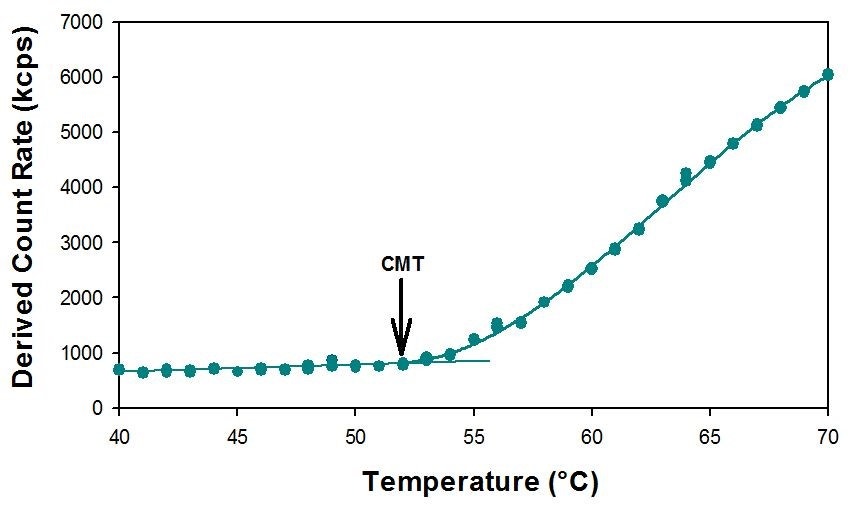
Figure 1 – Derived count rates for the sample F-68 (10 mg/mL) at different temperatures, showing an inflection point around 52°C – the temperature at which micelles start to be formed (CMT). This analysis was performed in triplicates.
With increasing concentration, it was noted that the CMT decreased by several degrees Celsius (Figure 2). This has been ascribed to the reduced hydration at higher concentrations and thus, at high temperatures, the increased hydrophobicity of the polymer molecules4.
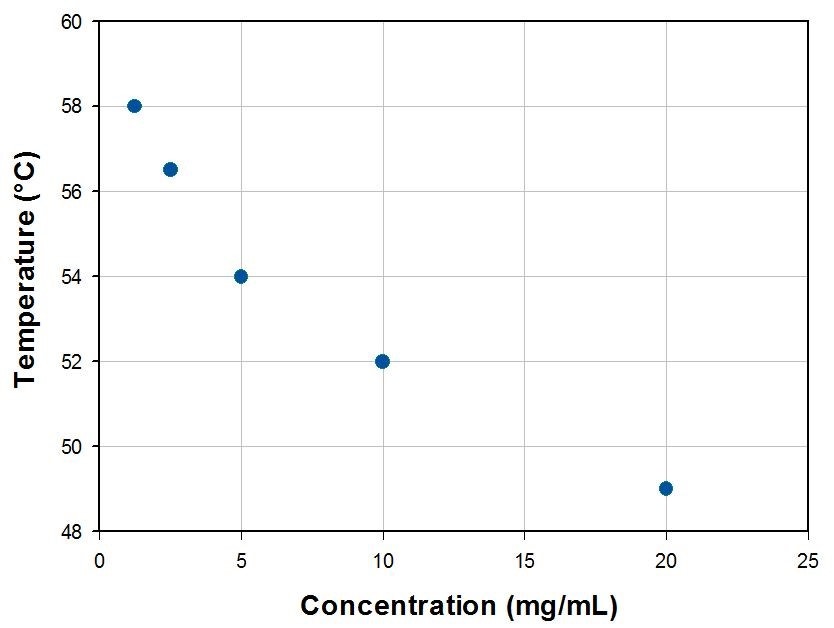
Figure 2 – Concentration dependence of the F-68’s CMT.
These samples were also run in parallel in a differential scanning calorimetry (DSC) instrument. Each CMT point obtained in DLS varied on average 0.5°C to those obtained in the DSC, showing the feasibility of the method described here.
Dynamic light scattering as a technique is known to produce low resolution results, particularly in the presence of similarly sized modes. The multi-angle dynamic light scattering (MADLS) feature in the Zetasizer Ultra, allows for a higher resolution size determination of multimodal samples, by using the three different angles (back, side and forward scattering detection) and combining the information obtained into one size distribution plot. This allowed for a better resolved size distribution at the CMT, where both unimers and micelles are present, as shown in Figure 3. Furthermore, when each angle was individually analyzed it was not possible to discriminate the modes for the unimer and micelle, proving that the MADLS analysis gives a higher resolution result (Figure 4).
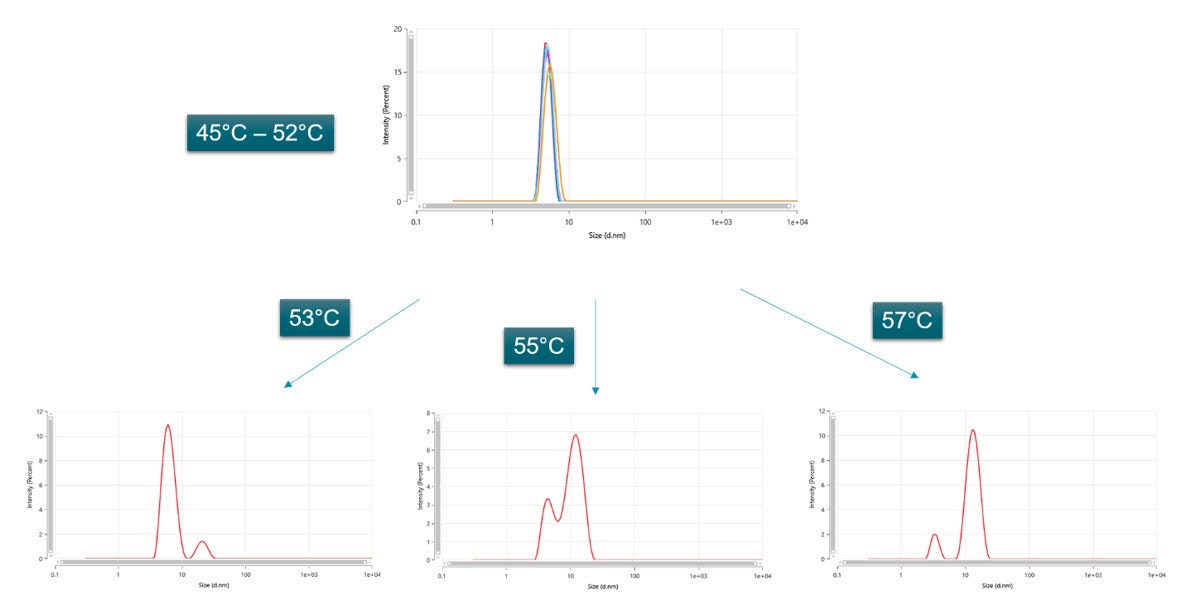
Figure 3 – Multi-angle dynamic light (MADLS) scattering result for the sample F-68 (10 mg/mL) at different temperatures, showing the transition from unimers to micelles.
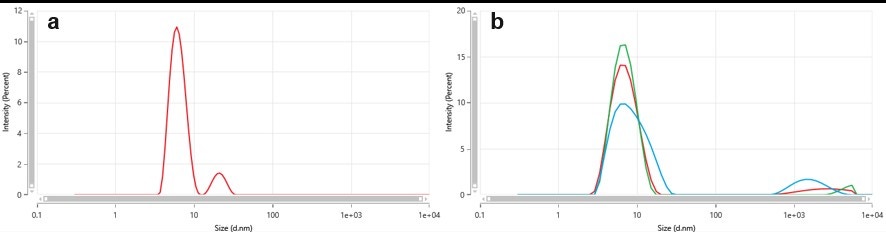
Figure 4 – Comparison between the MADLS result (a) and the 3 individual angle measurements (b) for the 10 mg/mL F-68 at 53°C (above CMT; red, green and blue colors represent back, side and forward detection angles, respectively).
It has been shown in this application note that the Zetasizer Ultra can be used to determine the critical micellization temperature of Pluronic surfactants. In contrast with previous light-scattering studies, the Zetasizer Ultra can significantly decrease the measurement time and simplify the experimental procedure. It does not require numerous sample filtrations and long data collection times. Moreover, the MADLS measurements allows for a higher resolution size distribution plot to be obtained which makes it possible for better monitoring of the transition stage between micelles and unimers.
1. Alexandridis, P. & Alan Hatton, T. Poly(ethylene oxide) poly(propylene oxide) poly(ethylene oxide) block copolymer surfactants in aqueous solutions and at interfaces: thermodynamics, structure, dynamics, and modeling. Colloids Surfaces A Physicochem. Eng. Asp. 96, 1–46 (1995).
2. Gaucher, G. et al. Block copolymer micelles: Preparation, characterization and application in drug delivery. J. Control. Release 109, 169–188 (2005).
3. Yeon, C., Lee, I., Kim, G. H. & Yun, S. J. Unimer-Assisted Exfoliation for Highly Concentrated Aqueous Dispersion Solutions of Single- and Few-Layered van der Waals Materials. Langmuir 33, 1217–1226 (2017).
4. Zhou, Z. K. & Chu, B. Light-Scattering Study on the Association Behavior of Triblock Polymers of Ethylene-Oxide and Propylene-Oxide in Aqueous-Solution. J. Colloid Interface Sci. 126, 171–180 (1988).
5. Matsuoka, H., Moriya, S. & Yusa, S. ichi. Fundamental properties, self-assembling behavior, and their temperature and salt responsivity of ionic amphiphilic diblock copolymer having poly(N-isopropylacrylamide) in aqueous solution. Colloid Polym. Sci. 296, 77–88 (2018)