Zeta potential, a measure of the magnitude of the repulsion or attraction between particles, is an important parameter across a diverse range of industries including brewing, ceramics, pharmaceuticals and water treatment. However, performing zeta potential measurements can be complicated and some expertise is required to obtain accurate and reliable results. With the new Zetasizer Nano series, Malvern Instruments has not only simplified zeta potential measurements, but also improved measurement accuracy with the implementation of a new measurement technology called M3-PALS.
Knowledge of the zeta potential of a dispersion gives the possibility of controlling the electrostatic interactions in the dispersion, and hence control the stability of the emulsion or dispersion. This can be critical to efficacy, shelf-life and product performance.
Over the past 20 years, as the importance of zeta potential has become recognized, the technology and instrumentation available have improved significantly. However, some issues remained, including the ease-of-use of the instrument and cross contamination of samples in the system itself.
To address these problems, the Zetasizer Nano system uses M3-PALS technology for zeta potential measurements. This is a second-generation implementation of the well-established technique of phase analysis light scattering (PALS) combined with the patented mixed-mode measurement (M3) technique. The combination of these techniques has resulted in simplified, more accurate measurements and has enabled the development of the world's first disposable cell for zeta potential measurements, eliminating cross contamination.
PALS is a variation of laser Doppler velocimetry (LDV). LDV uses a classical electrophoresis cell, with a potential applied, and measures the particle mobility generated using the frequency shift of light caused by particles moving in the cell. The mobility measured is converted to zeta potential using established theories. This technique requires the application of large fields that can result in thermal problems with highly conductive samples.
PALS is a much more sensitive technique as it uses the information contained in the phase of the scattered light to determine the frequency shift. This method uses the same optical setup as the LDV method, but a different signal processing method is employed. The measured phase shift is directly proportional to the change in position of the particles. Using the phase gives a resolution discrimination of around a thousand times greater than does frequency analysis.
The PALS technique was developed originally to improve measurement sensitivity for low mobility samples where the electrophoretic mobility is lower than the typical 10-8 m2/sV found in aqueous media. It has generally been implemented using a sinusoidal electric field across simple parallel plate electrodes, and the ability to discriminate electrophoretic motion from thermal drift is particularly useful in such an arrangement where temperature stability of the sample is hard to achieve due to Joule heating.
M3 is a relatively new technique for measuring zeta potential using electrophoresis in a capillary cell. It combines the best features of measurement at the stationary layer (described below) and the recently described fast field reversal (FFR) technique (also described below), to provide unprecedented accuracy and resolution.
When subjected to an electric field, micron sized particles, which have very low inertia, reach terminal velocity in microseconds. Actual velocity is determined by the charge associated with the particle (zeta potential), the viscosity of the medium and the applied field.
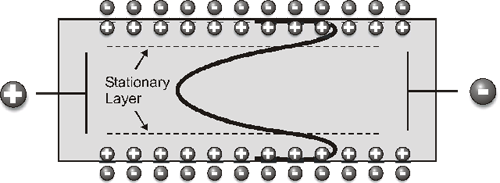
Capillary cells are the favored choice for microelectrophoresis as they have been demonstrated to give the highest resolution. However, if the capillary cell walls have a charge, this will give rise to a zeta potential at the wall and produces an effect called electro-osmosis when a potential is applied across the capillary. This effect causes the liquid to stream along the cell wall, and in a capillary cell that is closed at both ends, the liquid returns along the center of the cell. True particle mobility, from which zeta potential is derived, is measured at the stationary layer, the point where fluid flow along the cell walls cancels out the return flow in the center of the cell (Figure 1).
|
This layer must be precisely located and the cell must be clean. However, if electro-osmosis could be avoided, then the mobility of particles at any point in the cell would be the true mobility, alleviating the need to precisely locate a single point for taking measurements.
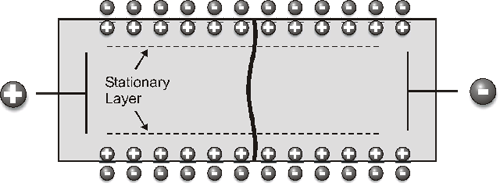
Under the usual microelectrophoresis conditions, a colloidal particle responds much faster to an applied electric field than does the liquid inside the capillary. Therefore, it is possible to apply the alternating electric field with a high enough frequency that electro-osmosis is suppressed, whereas the particles will follow the field with the same velocity as when a direct current is applied. This is the fast field reversal (FFR) technique and means the zeta potential measurement can be made at any position within the capillary cell (Figure 2)
|
This FFR technique does have a disadvantage in that the measurement period is so short due to the short application of the field, that information about the distribution is degraded, a factor addressed by the M3 technique.
M3 consists of taking two measurements for each zeta potential determination, one with the applied field being reversed slowly, and a second with a rapidly reversing applied field. This means it is no longer necessary for the operator to select any analysis parameters for the measurement, as the appropriate settings are calculated as part of the M3 sequence. The FFR part of the measurement gives accurately determines the mean, and the slow field application recovers the distribution information, combining the best features of both methods.
The robustness of M3-PALS makes it ideal for comparative studies over prolonged periods, for example in the measurement of pharmaceutical formulations for stability testing. Improved resolution also allows mixtures of partially coated and fully coated materials to be resolved, including coated latices for diagnostic kits. In addition, the high-resolution mode can be used to follow the interaction between materials with different zeta potentials such as DNA and liposomes for gene therapy studies.
The real benefit of M3-PALS is that it provides a strategy for accurately measuring the zeta potential of high and low mobility samples. Samples can have low mobility due to the low dielectric constant of the medium, a high sample viscosity or a high dispersant conductivity. Results are available as a distribution, rather than simply the mean value produced by a standard PALS measurement. This enables users to distinguish between a real change in the mean value of the peak of interest, and a change due to the appearance of populations at other zeta potentials.
The use of M3-PALS technology has made possible the development of a fully disposable zeta potential measurement cell (Figure 3).

|
Since the measurement position inside the cell is no longer absolutely critical, it has become possible to manufacture a unique, disposable polycarbonate cell, that consists of gold plated electrodes and a folded capillary cell molded into a single measurement chamber. The cell can be used repeatedly, or discarded after a single use and is central to the concept of simple zeta potential measurement in this measurement system.
The combination of M3 and PALS technology represents the next generation in zeta potential measurement. Not only is cross-contamination eliminated, the technique allows operators with minimal training to obtain accurate, repeatable measurements over a wide range of applications.