






PEAQ-DSC Automated 是一種 MicroCal 技術,系統透過自動化的整合式平台,以較低的樣品耗用量,執行大量的高靈敏度蛋白質穩定性分析,可提高生產力。 所有的槽填充與清潔功能均為全自動,無須額外人為操作。
選擇型號:
尋找更多資訊?
如需報價、更多資訊或下載手冊,請選擇下面的選項。
高處理量、高靈敏度的 PEAQ-DSC Automated 是一種 MicroCal 技術,系統軟體,能透過簡單的實驗設定和彈性的儀器排程來簡化工作流程。
自動化數據分析功能可支援產生高完整性的熱穩定性數據,並能遵循法規要求,同時還可與現有的數據處理和傳輸系統輕鬆整合。
PEAQ-DSC 也可作為無自動取樣器的手動系統使用。

判別穩定性的黃金標準,且僅需極少的檢定開發
以直接、免標記的方式測量溶液中生物分子的狀態穩定性
標準 96 孔槽盤形式提供大容量,裝載容易,並具備可容納高達 6 個槽盤的恆溫控制儲存區
完全整合式自動取樣器每日可進行多達 50 個樣品的無人分析
具備自動槽填充與槽清潔功能,可在無人值守的情況下運作
可測量極強的結合常數,最高可達 1020M-1
功能強大的 MicroCal PEAQ-DSC 軟體大幅縮短一般數據的分析時間

MicroCal PEAQ-DSC Automated 所獲取的數據可在生物製藥開發中提供關鍵性的指引,協助進行蛋白質工程、(預) 配方開發、製程開發、製造變更控制,以及生物相似性和生物可比較性研究。
無須人力看管的操作系統可進行 24 小時的篩選作業,其整合式的軟體也能簡化工作流程,進而提供客觀數據分析和效能鑑定,並能符合 21 CFR Part 11 和附錄 11 的法規,各方面都能提供高完整性的數據,並推動生物製藥研究的生產力。
功能強大的 MicroCal PEAQ-DSC 軟體大幅縮短一般數據的分析時間,同時還包括:
差示掃描熱分析儀 (DSC) 是功能強大的分析工具,能分析蛋白質和其他生物分子的熱穩定性,此技術可測量溶液中分子因熱導致結構轉變所產生的焓 (ΔH) 和溫度 (Tm) 變化,這些資訊對於了解蛋白質、核酸、微胞錯合物和其他高分子系統的穩定或去穩定因子,可提供寶貴的見解和數據,而這些數據可用來預測生物分子產品 (含生物製藥) 的有效期限、進行批次對批次及生物相似性分子對新穎分子的比較、研擬純化策略、分析和評估蛋白質構造特性,以及在小分子藥物探索計畫中排序配體對其蛋白質標的親和力。
數據分析及測量/衍生參數在示差掃描熱分析儀技術頁面中有進一步的詳細說明。
蛋白質和其他高分子形成的功能性結構經常經歷由溫度誘發的構形變化,例如蛋白質展開,這些變化會因非共價鍵在分子內重新分配排列而造成熱量的吸收,示差掃描熱分析儀可以非常精確地測量此一熱量吸收。
MicroCal PEAQ-DSC 系統的熱分析核心包含一樣品槽 (含待測樣品) 和參考槽 (含相應的緩衝溶液),兩者均置於絕緣護套中,這兩個測試槽始終維持於相同溫度,並在測量期間以恆定的掃描速率加熱。
當樣品槽內的分子發生結構展開時會吸收熱量,在樣品槽和參考槽之間產生溫度差 (ΔT),該溫度差在 Peltier 模組的兩端產生熱梯度,進而生成相應比例的電壓,此電壓會依序轉換為功率,在 Peltier 模組形成反饋迴路,以使 ΔT 回歸為零。
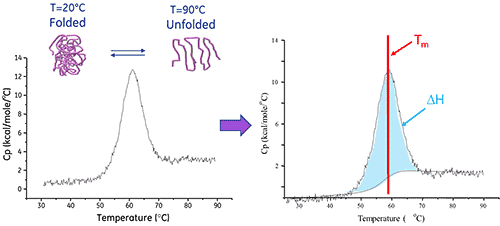
由於蛋白質展開是吸熱過程,因此在熱分析圖中會觀察到正位移,此一蛋白質「熔融」轉變的中點為 Tm,而曲線下的面積等於展開過程中的焓變化 (ΔH) (請參閱下圖)。
PEAQ-DSC 自動化系統在整個生物製藥行業中廣受信賴,可為生物治療藥物在發現、開發和生產階段提供有價值的穩定性數據。這是由於該檢測方法的通用性、能夠檢測出生物藥物構象的極其微小變化,以及數據的高重複性。
網路研討會: 使用 DSC 解決預配方與製程開發中的穩定性問題
MicroCal PEAQ-DSC 儀器的高度可重複性,以及該技術能夠檢測出蛋白質中哪怕一個胺基酸的變化,這些都是 DSC 數據在生物相似藥核准申請中被廣泛使用的核心原因。MicroCal PEAQ-DSC 軟體配備了用於與原研藥進行統計比較的生物相似性工具。
DSC、DLS 及其他生物物理技術可用於研究抗體偶聯藥物候選物的穩定性,以及抗體與藥物比例對其穩定性與聚集傾向的影響。以下示例亦展示了如何利用 DSC 在廣泛的掃描速率範圍內測量加熱失活的動力學。
網路研討會:抗體偶聯藥物的生物物理與穩定性表徵
| 技术类型 | 示差掃描熱分析儀 |
|---|---|
| 测量类型 | 溫度中點 (TM)
焓 (ΔH)
熱容變化 (ΔCp)
|
| 样品池 | 毛細管 |
|---|---|
| 样品池材料 | 鉭 |
| 样品池容积 | 130 μL |
| 样品容量 | 288 (6 個 96 孔槽盤) |
|---|---|
| 样品量 | 325 µL |
| 典型样本浓度 | 0.01 mg/mL – 10 mg/mL 1 |
| 样品处理量 | ≤50 個樣品/天 |
| 样品储存温度范围 | 4 - 40 °C |
| 噪音 | 0.05 μCal/°C 2 |
|---|---|
| 基线可重复性 | 1 μCal/°C 2 |
| 响应时间 | 5s 2 |
| 重複性 | <0.2 µCal/°C 3 |
| 再現性 | <0.08°C 標準差TM 和 ΔH <2% RSD4 |
| 系统可重复性 | <0.1°C 標準差TM 和 ΔH <5% RSD4 |
| 多重反馈模式 | 有 (被動式、高增益和低增益) |
| 温度范围 | 2°C 至 130°C 2, 5 |
| 最大扫描速率 | 240 °C/h |
| 逆向扫描 | 是 |
| 压力扰动量热法 (PPC) | N/A |
| 清洗程序 | 3 種預先程式設定的例行程序 |
| 清洗溶剂 | 依標準使用水和清潔劑 |
| 21 CFR 第 11 部分 | 有,具備 PEAQ-Compliance 選配軟體 |
|---|---|
| 已连接网络 | 有,具備電子郵件提醒功能 |
| 操作溫度 | 10 - 28 °C |
|---|---|
| 储存温度 | -20 - 50 °C |
| 濕度 | 10% 至 70%,無冷凝 (針對儲存而言為 10% 至 90%) |
| 防護等級(IP) | IP21 |
| 電功率 | 100-240 V A/C,50/60 Hz,70 W (槽),400 W (最大,自動取樣器),PC 依照其需求 |
| 认证 | CE (EN61010-1), EMC (EN61326-2-1, EN61326-1, FCC, ICES, VCCI), ISO9001:2008 |
| 尺寸 (宽, 长, 高): | 101 cm × 68 cm × 70 cm |
|---|---|
| 重量 | 25 kg |
1 依樣品而定
2 在 60°C/h 的高反饋下,核糖核酸酶 (RNase) 在 pH 值 5.5 的 50 mM KAc 緩衝液中量測的典型結果
3 重新掃描穩定緩衝液
4 使用核糖核酸酶 (RNase)
5 範圍可依要求向下延伸至 -10 °C
|
版本号: MAN0590-02-EN-00
版本号: MAN0589-02-EN-00
MicroCal PEAQ-DSC Software Update v1.65 - Maintenance release
MicroCal PEAQ-DSC Software Update v1.64 - Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Initial release of MicroCal PEAQ-DSC software
透過 Malvern Panalytical 的服務計畫保護您的投資並確保機臺隨時都具有最佳效能。
我們瞭解當您購買 Malvern Panalytical 的產品時,這只是雙方合作關係的第一階段,其關係將持續到遺棄的整個使用壽命。因此根據您的需求, Malvern Panalytical 將為您量身打造後續的設備維護方案
對實驗室來說,最大化遺棄的正常運行時間對其日常效能至關重要。若是不幸發生,我們最高等級的回應與「全權負責」的價格計畫將會處理那些意外的維修費用。
藉由維持儀器的效率以提高實驗室的生產力。仍然具備優先回應待遇,再配合專業技術與軟體的支援,我們將維持您的儀器效能能夠正常運作。
對於期望優化儀器效能、定期預防性維護至關重要。
| Platinum Plan | Gold Plan | Bronze Visit | |
|---|---|---|---|
| 年度 PM/PV 測試 |
有
|
有
|
無
|
| 電話/e-mail 支援 |
有
|
有
|
無
|
| 優先回應 |
有
|
有
|
無
|
| 緊急故障叫修* |
有
|
有
|
無
|
| 包含零件 |
有
|
無
|
無
|
| 軟體和技術支援 |
有
|
有
|
有
|
| IQ/OQ (製藥業)*** |
有
|
有
|
有
|
*包含維修和差旅費用 ***需支付額外費用
高處理量、高靈敏度的 PEAQ-DSC Automated 是一種 MicroCal 技術,系統軟體,能透過簡單的實驗設定和彈性的儀器排程來簡化工作流程。
自動化數據分析功能可支援產生高完整性的熱穩定性數據,並能遵循法規要求,同時還可與現有的數據處理和傳輸系統輕鬆整合。
PEAQ-DSC 也可作為無自動取樣器的手動系統使用。

判別穩定性的黃金標準,且僅需極少的檢定開發
以直接、免標記的方式測量溶液中生物分子的狀態穩定性
標準 96 孔槽盤形式提供大容量,裝載容易,並具備可容納高達 6 個槽盤的恆溫控制儲存區
完全整合式自動取樣器每日可進行多達 50 個樣品的無人分析
具備自動槽填充與槽清潔功能,可在無人值守的情況下運作
可測量極強的結合常數,最高可達 1020M-1
功能強大的 MicroCal PEAQ-DSC 軟體大幅縮短一般數據的分析時間

MicroCal PEAQ-DSC Automated 所獲取的數據可在生物製藥開發中提供關鍵性的指引,協助進行蛋白質工程、(預) 配方開發、製程開發、製造變更控制,以及生物相似性和生物可比較性研究。
無須人力看管的操作系統可進行 24 小時的篩選作業,其整合式的軟體也能簡化工作流程,進而提供客觀數據分析和效能鑑定,並能符合 21 CFR Part 11 和附錄 11 的法規,各方面都能提供高完整性的數據,並推動生物製藥研究的生產力。
功能強大的 MicroCal PEAQ-DSC 軟體大幅縮短一般數據的分析時間,同時還包括:

差示掃描熱分析儀 (DSC) 是功能強大的分析工具,能分析蛋白質和其他生物分子的熱穩定性,此技術可測量溶液中分子因熱導致結構轉變所產生的焓 (ΔH) 和溫度 (Tm) 變化,這些資訊對於了解蛋白質、核酸、微胞錯合物和其他高分子系統的穩定或去穩定因子,可提供寶貴的見解和數據,而這些數據可用來預測生物分子產品 (含生物製藥) 的有效期限、進行批次對批次及生物相似性分子對新穎分子的比較、研擬純化策略、分析和評估蛋白質構造特性,以及在小分子藥物探索計畫中排序配體對其蛋白質標的親和力。
數據分析及測量/衍生參數在示差掃描熱分析儀技術頁面中有進一步的詳細說明。
蛋白質和其他高分子形成的功能性結構經常經歷由溫度誘發的構形變化,例如蛋白質展開,這些變化會因非共價鍵在分子內重新分配排列而造成熱量的吸收,示差掃描熱分析儀可以非常精確地測量此一熱量吸收。
MicroCal PEAQ-DSC 系統的熱分析核心包含一樣品槽 (含待測樣品) 和參考槽 (含相應的緩衝溶液),兩者均置於絕緣護套中,這兩個測試槽始終維持於相同溫度,並在測量期間以恆定的掃描速率加熱。
當樣品槽內的分子發生結構展開時會吸收熱量,在樣品槽和參考槽之間產生溫度差 (ΔT),該溫度差在 Peltier 模組的兩端產生熱梯度,進而生成相應比例的電壓,此電壓會依序轉換為功率,在 Peltier 模組形成反饋迴路,以使 ΔT 回歸為零。
由於蛋白質展開是吸熱過程,因此在熱分析圖中會觀察到正位移,此一蛋白質「熔融」轉變的中點為 Tm,而曲線下的面積等於展開過程中的焓變化 (ΔH) (請參閱下圖)。

PEAQ-DSC 自動化系統在整個生物製藥行業中廣受信賴,可為生物治療藥物在發現、開發和生產階段提供有價值的穩定性數據。這是由於該檢測方法的通用性、能夠檢測出生物藥物構象的極其微小變化,以及數據的高重複性。
網路研討會: 使用 DSC 解決預配方與製程開發中的穩定性問題
MicroCal PEAQ-DSC 儀器的高度可重複性,以及該技術能夠檢測出蛋白質中哪怕一個胺基酸的變化,這些都是 DSC 數據在生物相似藥核准申請中被廣泛使用的核心原因。MicroCal PEAQ-DSC 軟體配備了用於與原研藥進行統計比較的生物相似性工具。
DSC、DLS 及其他生物物理技術可用於研究抗體偶聯藥物候選物的穩定性,以及抗體與藥物比例對其穩定性與聚集傾向的影響。以下示例亦展示了如何利用 DSC 在廣泛的掃描速率範圍內測量加熱失活的動力學。
網路研討會:抗體偶聯藥物的生物物理與穩定性表徵
| 技术类型 | 示差掃描熱分析儀 |
|---|---|
| 测量类型 | 溫度中點 (TM)
焓 (ΔH)
熱容變化 (ΔCp)
|
| 样品池 | 毛細管 |
|---|---|
| 样品池材料 | 鉭 |
| 样品池容积 | 130 μL |
| 样品容量 | 288 (6 個 96 孔槽盤) |
|---|---|
| 样品量 | 325 µL |
| 典型样本浓度 | 0.01 mg/mL – 10 mg/mL 1 |
| 样品处理量 | ≤50 個樣品/天 |
| 样品储存温度范围 | 4 - 40 °C |
| 噪音 | 0.05 μCal/°C 2 |
|---|---|
| 基线可重复性 | 1 μCal/°C 2 |
| 响应时间 | 5s 2 |
| 重複性 | <0.2 µCal/°C 3 |
| 再現性 | <0.08°C 標準差TM 和 ΔH <2% RSD4 |
| 系统可重复性 | <0.1°C 標準差TM 和 ΔH <5% RSD4 |
| 多重反馈模式 | 有 (被動式、高增益和低增益) |
| 温度范围 | 2°C 至 130°C 2, 5 |
| 最大扫描速率 | 240 °C/h |
| 逆向扫描 | 是 |
| 压力扰动量热法 (PPC) | N/A |
| 清洗程序 | 3 種預先程式設定的例行程序 |
| 清洗溶剂 | 依標準使用水和清潔劑 |
| 21 CFR 第 11 部分 | 有,具備 PEAQ-Compliance 選配軟體 |
|---|---|
| 已连接网络 | 有,具備電子郵件提醒功能 |
| 操作溫度 | 10 - 28 °C |
|---|---|
| 储存温度 | -20 - 50 °C |
| 濕度 | 10% 至 70%,無冷凝 (針對儲存而言為 10% 至 90%) |
| 防護等級(IP) | IP21 |
| 電功率 | 100-240 V A/C,50/60 Hz,70 W (槽),400 W (最大,自動取樣器),PC 依照其需求 |
| 认证 | CE (EN61010-1), EMC (EN61326-2-1, EN61326-1, FCC, ICES, VCCI), ISO9001:2008 |
| 尺寸 (宽, 长, 高): | 101 cm × 68 cm × 70 cm |
|---|---|
| 重量 | 25 kg |
1 依樣品而定
2 在 60°C/h 的高反饋下,核糖核酸酶 (RNase) 在 pH 值 5.5 的 50 mM KAc 緩衝液中量測的典型結果
3 重新掃描穩定緩衝液
4 使用核糖核酸酶 (RNase)
5 範圍可依要求向下延伸至 -10 °C
|
版本号: MAN0590-02-EN-00
版本号: MAN0589-02-EN-00
MicroCal PEAQ-DSC Software Update v1.65 - Maintenance release
MicroCal PEAQ-DSC Software Update v1.64 - Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Maintenance release
Initial release of MicroCal PEAQ-DSC software
透過 Malvern Panalytical 的服務計畫保護您的投資並確保機臺隨時都具有最佳效能。
我們瞭解當您購買 Malvern Panalytical 的產品時,這只是雙方合作關係的第一階段,其關係將持續到遺棄的整個使用壽命。因此根據您的需求, Malvern Panalytical 將為您量身打造後續的設備維護方案
對實驗室來說,最大化遺棄的正常運行時間對其日常效能至關重要。若是不幸發生,我們最高等級的回應與「全權負責」的價格計畫將會處理那些意外的維修費用。
藉由維持儀器的效率以提高實驗室的生產力。仍然具備優先回應待遇,再配合專業技術與軟體的支援,我們將維持您的儀器效能能夠正常運作。
對於期望優化儀器效能、定期預防性維護至關重要。
| Platinum Plan | Gold Plan | Bronze Visit | |
|---|---|---|---|
| 年度 PM/PV 測試 |
有
|
有
|
無
|
| 電話/e-mail 支援 |
有
|
有
|
無
|
| 優先回應 |
有
|
有
|
無
|
| 緊急故障叫修* |
有
|
有
|
無
|
| 包含零件 |
有
|
無
|
無
|
| 軟體和技術支援 |
有
|
有
|
有
|
| IQ/OQ (製藥業)*** |
有
|
有
|
有
|
*包含維修和差旅費用 ***需支付額外費用