Grating-coupled interferometry (GCI)
Benefit from the revolution in the studies of biomolecular interactions
The Kinetics Guide | Binding kinetics with the WAVE system. Download now
Download nowBenefit from the revolution in the studies of biomolecular interactions

Engineered around a proprietary Grating-Coupled Interferometry (GCI) technology to deliver improved data quality from label-free biomolecular interaction analysis, the WAVEsystem builds on Waveguide Interferometry to achieve superior resolution in signal and time compared to traditional Surface Plasmon Resonance. This allows researchers to quickly and accurately measure kinetic rates, determine affinity constants, and monitor the concentrations of even low abundance interacting analytes in crude samples such as biofluids. With unrivaled flexibility and high sensitivity, the WAVE brings label-free quantification to a whole new world of applications, revolutionizing the study of biomolecular interactions.
GCI is our cutting-edge biophysical characterization method commercially available since 2015 in the WAVE family of laboratory devices.
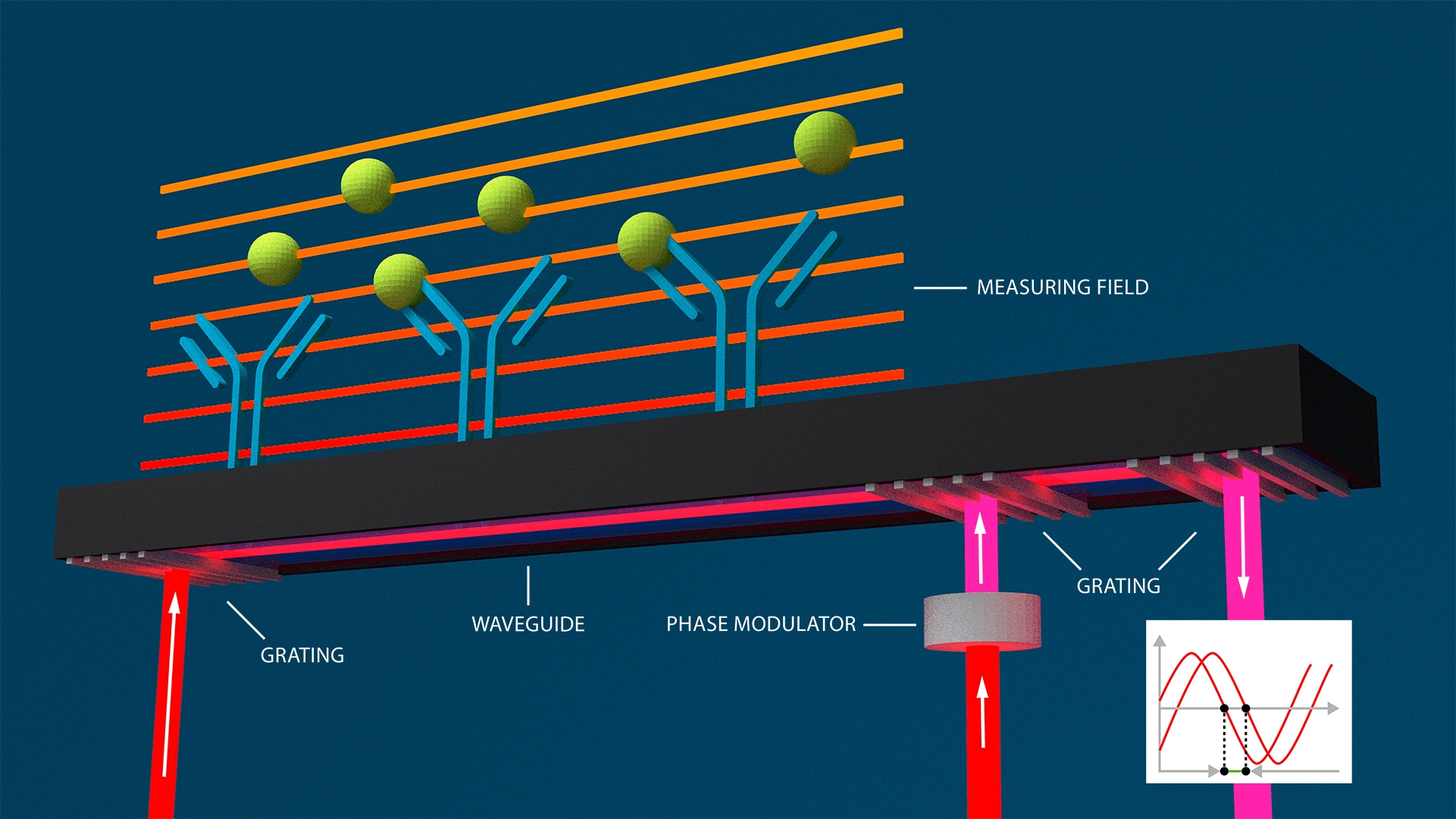
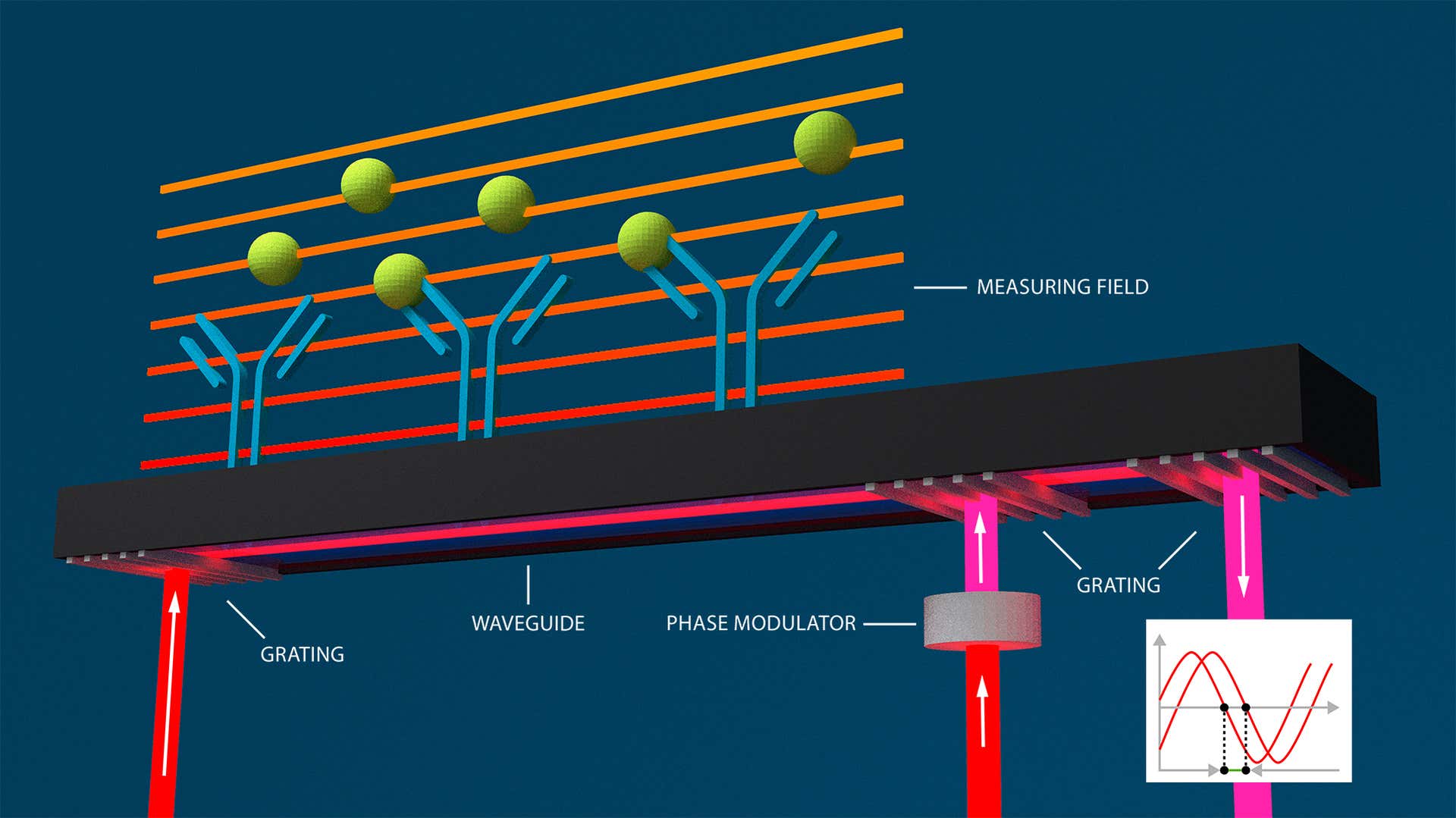
Our patented Grating-Coupled Interferometry design leverages and enhances the intrinsic benefits of Waveguide Interferometry to exceed the sensitivity levels of Surface Plasmon Resonance. Like Waveguide Interferometry, the evanescent field penetrates less deep into the sample and extends the light-to-sample interaction length for improved signal-to-noise ratios (<0.01 pg/mm2).
However, the Creoptix GCI readout scheme has the advantage that the interferogram is created in the time domain and within the waveguide, instead of being projected onto a CCD camera. Measuring refractive index changes on the sensor surface as time-dependent phase-shift signals therefore provides a more robust readout compared to classical Waveguide Interferometry or Surface Plasmon Resonance, regardless of temperature drifts or vibrations, translating to superior resolution in signal and time.
| Grating-Coupled Interferometry (GCI) | Surface Plasmon Resonance (SPR) | Biolayer Interferometry (BLI) | |
|---|---|---|---|
|
Broadest application range
Suitable for a variety of molecules ranging from low to high molecular weights, purified or crude. |
Yes
Suitable for Fragments, Small Molecules, Peptides, Proteins, Viruses, Cell Culture Supernatants, Serums, Cell lysates |
No
Suitable for Small Molecules, Peptides (limited suitability for Fragments, Viruses, Cell Culture Supernatants, Serums, Cell lysates) |
No
Suitable for Cell Culture Supernatants, Serums, Cell lysates (limited suitability for Peptides, Proteins, Viruses) |
|
Measure weakest binders
Ability to measure kinetics with fast off-rates thanks to fast fluidics and high acquisition rates. |
Yes
Off-rates up to kd=10 s-1 |
No
Off-rates up to kd=1 s-1 |
No
Off-rates up to kd=0.1 s-1 |
|
Measure tightest binders
Ability to accurately measure kinetics even for tight binders and fast on-rates. |
Yes
Measurement under flow conditions |
Yes
Measurement under flow conditions |
No
Measurement under diffusion-limited conditions (no microfluidics) |
|
Low system maintenance
Little downtime due to service or unexpected repairs. |
Yes
No-clog microfluidics |
No
Traditional microfluidics |
Yes
No microfluidics |
Molecular interactions are detected as changes in refractive index within an evanescent field (orange) causing a phase shift of the beam in the waveguide and hence an interference to a reference beam projected in parallel to a screen.