

Solutions d'analyse physicochimique et expertise pour faciliter le développement de formulations de médicaments, conformément aux exigences en matière de biodisponibilité et de traitement

Le développement de formulations pharmaceutiques associe la découverte d'une nouvelle substance médicamenteuse au développement réussi d'un médicament commercial. Les chercheurs spécialisés dans le développement de formulations doivent déterminer la voie la plus appropriée pour parvenir à une administration efficace du médicament en fonction des besoins du patient, mais également optimiser les caractéristiques de la formulation en fonction d'une connaissance des exigences de biodisponibilité et de traitement du médicament.
Il s'agit d'un défi majeur. Seuls 10 % des nouveaux médicaments dont les formulations pré-cliniques sont en cours de développement sont commercialisés. Les coûts de développement pharmaceutique et la pression pour libérer la valeur du circuit du médicament étant en hausse, les sociétés pharmaceutiques innovantes font des efforts significatifs pour déterminer comment accélérer la sélection efficace des formulations. Il est essentiel de développer une compréhension complète de la forme et de la structure de la substance médicamenteuse et du médicament.
Les sociétés pharmaceutiques de médicaments génériques connaissent des défis similaires, le développement réussi de formulations de médicaments complexes restant difficile. Il existe un marché non exploité important pour les nouveaux génériques complexes. Mise en avant par les organismes internationaux de réglementation, il est désormais reconnu qu'une compréhension physicochimique détaillée de la microstructure des formes de dosage complexes peut permettre un développement de produits réussi.
Les techniques physicochimiques de Malvern Panalytical sont régulièrement appliquées pour relever ces défis de formulation. Nos solutions sont utilisées pour caractériser les propriétés physicochimiques et mieux comprendre la substance médicamenteuse ainsi que la forme du médicament, dans le but de faciliter la sélection des excipients et de comprendre leur fonction dans les formulations, mais également d'évaluer la manière dont le stockage ou le traitement peuvent altérer les performances du médicament. Notre expertise permet de fournir les bonnes données en proposant des informations afin de réussir le développement des formulations.





Le développement d'une formulation pharmaceutique réussie requiert l'association du principe actif pharmaceutique (API) et d'excipients inactifs. Les excipients peuvent être des agents de gonflement simples, conçus pour favoriser le contrôle de l'uniformité du contenu de la dose. Cependant, certains excipients jouent de plus en plus un rôle fonctionnel dans le contrôle de la libération du médicament ou dans la garantie que le médicament atteindra la zone d'action souhaitée. Dans ce cas, la compatibilité entre l'excipient sélectionné et la substance médicamenteuse est essentielle pour s'assurer que la bonne dose est libérée dans la fenêtre thérapeutique requise. L'analyse physicochimique peut faciliter la sélection d'excipients, permettre la stabilité de la substance médicamenteuse et du médicament à évaluer, mais également garantir que les attributs critiques des matériaux (CMA) liés aux performances de la formulation sont identifiés comme faisant partie de la définition de l'espace de conception appliqué aux contrôles de fabrication en aval.
Zetasizer Ultra permet de caractériser la stabilité et la qualité des dispersions, des émulsions et des crèmes, réduisant ainsi le temps de formulation et accélérant la commercialisation de nouveaux produits.
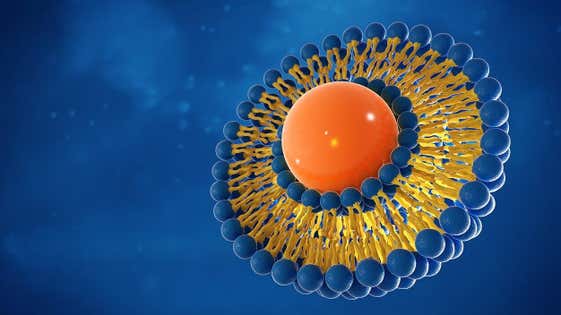
Les exigences de plus en plus complexes pour parvenir à une délivrance reproductible de médicaments constituent un défi commun pour les chercheurs en développement de formulations. De nombreux nouveaux principes actifs pharmaceutiques (API) sont peu solubles, ce qui signifie que l'administration de doses solides par voie orale traditionnelles n'est plus pertinente. La formulation est donc de plus en plus complexe, que ce soit pour permettre une meilleure biodisponibilité pour l'administration orale ou favoriser une administration locale, de façon à ce que la concentration du médicament sur la zone d'action réponde aux exigences thérapeutiques.
De nouveaux systèmes d'administration de médicaments, basés sur des liposomes ou d'autres systèmes d'administration de nanoparticules, sont utilisés plus fréquemment pour améliorer le ciblage des médicaments. La gamme de techniques d'analyse complémentaires de Malvern Panalytical permet aux développeurs de formulations de comprendre la formulation et la stabilité de l'API et des excipients. Cela favorise l'optimisation des formulations complexes, ce qui permet de gagner du temps dans le choix d'une formulation candidate efficace.
Les défis liés au développement de formulations complexes concernent également le développement de médicaments génériques. Les organismes de réglementation du monde entier ont reconnu les effets du manque de lancement de génériques complexes sur les coûts des soins de santé. Face à ce problème, ils ont publié des directives spécifiques au produit mettant en évidence le rôle de l'évaluation de l'équivalence physicochimique, ou Q3, dans le cadre de l'évaluation de la bioéquivalence d'un générique de test par rapport à un médicament générique approuvé (RLD).
L'application d'une approche de test de bioéquivalence in vitro pourrait réduire considérablement le délai de commercialisation de nouveaux génériques en éliminant le besoin de mener des études cliniques finales. La boîte à outils de techniques d'analyse physicochimique et d'expertise de Malvern Panalytical, permettant d'évaluer les propriétés du médicament et de sa formulation, a un rôle essentiel dans la réussite des études de bioéquivalence in vitro.

Le développement d'une formulation réussie de générique commence par la compréhension de la structure et des performances du médicament générique approuvé (RLD). Dans ce cas, comme pour les évaluations de bioéquivalence in vitro, l'analyse physicochimique joue un rôle important pour faire progresser la compréhension des exigences en matière de formulation pharmaceutique. La caractérisation proactive, quantitative, structurelle et morphologique de l'API et des excipients présents dans le produit RLD peut créer des prototypes d'optimisation de la formulation et réduire considérablement les risques liés au développement.
Les avantages de cette approche de déformulation ne se limitent pas aux sociétés de production de génériques. Des méthodes similaires sont également appliquées dans le développement et la fabrication de nouveaux médicaments, fournissant des informations pour aider à identifier les causes des changements dans les performances de la formulation au cours de l'intensification. Cela peut également aider les entreprises à comprendre les effets des changements post-commercialisation sur le procédé de fabrication ou le lieu de fabrication sur la performance d'un médicament.
Morphologi 4-ID peut être utilisé pour simplifier, mais aussi relever les difficultés de déformulation et élaborer une bioéquivalence in vitro. Elles peuvent également permettre de détecter les anomalies et les contaminants, et d’identifier les déviations du processus au cours de la fabrication.