Antibodies are the body's natural defence against bacteria, viruses, and other blood borne foreign substances. Because of their specificity and affinity to unique antigen determinants, antibodies are routinely used as active ingredients in pharmaceutical products, some examples being products for the treatment of sexually transmitted diseases and cancer, and anti-rejection formulations for bone marrow and organ transplants. Antibodies are proteins, and as such, are prone to aggregation under perturbating solution conditions. Because of the sensitivity to small amounts of aggregates, light scattering is ideally
suited for characterization of antibody
formulations.
|
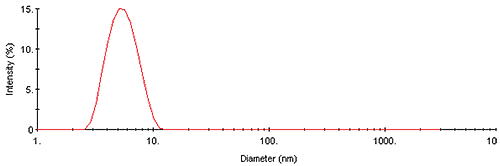
The Malvern Zetasizer Nano ZS system combines dynamic, static, and electrophoretic light scattering techniques in a single instrument, allowing the user to characterize their protein samples by size, mass, and charge. Figure 1 shows the size distribution, measured with a Malvern Zetasizer Nano ZS system, of a sample containing a monoclonal antibody fragment. As evident in this figure, the antibody fragment is non-aggregating under the current solution conditions and sample concentration, with the size distribution indicating a single particle family, with a Zavg diameter of 5.1nm.
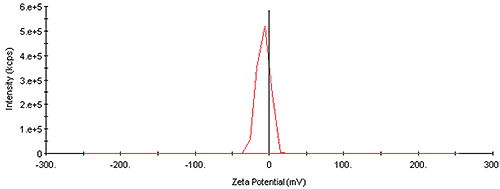
The zeta potential is representative of the particle charge, and is another common property used for monitoring formulation stability. The general rule of thumb is that particle formulations are more stable if the measured zeta potential is outside the range of +/- 20mV. The zeta distribution for the monoclonal antibody fragment is shown in Figure 2, which indicates a single particle distribution, with a zeta potential of -7.6mV. The relatively small value of the zeta potential under the current solution conditions suggests that the sample would be unstable at higher
concentrations.
|
The absolute molecular weight and 2nd virial coefficient of protein samples can also be measured with the Malvern Zetasizer Nano ZS. These properties are determined using a Debye plot, constructed from the concentration dependence of the sample scattering intensity. The Rayleigh expression used to construct a Debye plot is given in Equation 1, where K is an optical constant, C is the particle concentration, Rθ is the Rayleigh ratio of scattered to incident light intensity, M is the weight average particle molecular weight, and A2 is the 2nd virial coefficient.
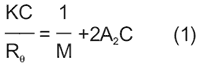
|
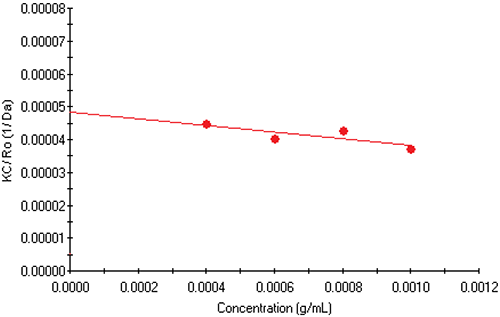
The Debye plot for the monoclonal antibody fragment examined here is shown in Figure 3. From the intercept and slope, the molecular weight of the antibody is 20.71 kDa and the 2nd virial
coefficient is -0.00499 mL mol / g2.
|
The measured molecular weight of 20.71kDa is consistent with the size exclusion chromatography estimated molecular weight of 18kDa for the sample. The 2nd virial coefficient is representative of the strength of particlesolvent interactions. Positive 2nd virial coefficient values indicate highly solvated particles, while negative values indicates low solubility. The value of -0.00499mL mol / g2 measured here indicates a low solubility under the current solutions conditions, and suggests that the formulation would be unstable at higher sample concentrations. These results are consistent with those from the zeta
measurements.
The Zetasizer Nano ZS system from Malvern Instruments is the first commercial instrument to include the hardware and software for combined dynamic, static, and electrophoretic light scattering measurements, giving the researcher a wide range of sample properties, including the size, molecular weight, and zeta potential. The system was specifically designed to meet the low concentration and sample volume requirements typically associated with pharmaceutical and biomolecular applications, along with the high concentration requirements for colloidal applications. Satisfying this unique mix of requirements was accomplished via the integration of a backscatter optical system and the design of a novel cell chamber. As a consequence of these features, the Zetasizer Nano ZS specifications for sample size and concentration exceed those for any other commercially available DLS instrument, with a size range of 0.6nm to 6um, and a concentration range of 0.1mg/mL lysozyme to 40% w/v. As an added bonus, the Zetasizer Nano ZS hardware is self optimizing, and the software includes a unique "one click" measure, analyze, and report feature designed to minimize the new user learning curve.