解離定数
解離定数(KD)とは、分子間相互作用および2つ以上の分子間の結合反応を測定するために使用される、動態パラメータです。
解離定数(KD)とは、分子間相互作用および2つ以上の分子間の結合反応を測定するために使用される、動態パラメータです。
このページでは、主に解離定数の計算方法や求め方を説明しています。結合親和性やKD値に関する概要は、以下のページをご参照ください。
解離定数KDは、分子間相互作用および2つ以上の分子間の結合反応を測定するために使用されるパラメータです。
KD値は、以下の式で表されます。
KD = [A][B] / [AB]
[A]:自由な分子Aの濃度
[B]:自由な分子Bの濃度
[AB]:結合した分子ABの濃度
解離定数はカイネティクス解析からも得ることができます。カイネティクス解析で求められる結合速度定数(ka)は、ある分子が別の分子と結合して複合体を形成する速度を表し、単位はM・S-1です。解離速度定数(kd)は、複合体が解離する速度を表し、単位はS-1です。解離定数KDは kd / kaで求められます。
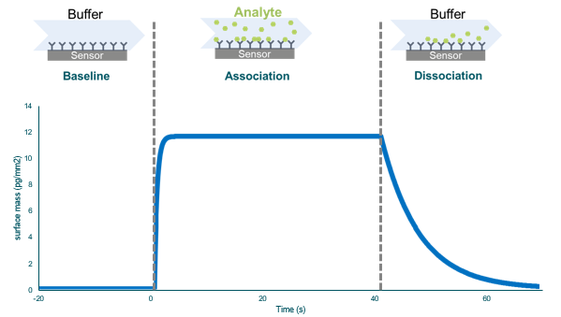
右のグラフは、分子間相互作用の測定結果を示したセンサーグラムです。まず、緩衝液をリガンドを固定化しているセンサー表面に流すことで、ベースラインが確立します。この後、サンプル(アナライト)が通過します。アナライトは、結合フェーズでリガンドと結合し、そこから結合速度定数kaを計算することができます。流れをサンプルから緩衝液へ戻すと、解離フェーズが始まります。
以下の図は2つの相互作用は同じ親和性(KD)を持っていますが、反応速度(kaとkd)は大きく異なります。
青線で表される相互作用は、結合が速く、解離も速いのに対し、赤線で表される相互作用は、結合がかなり遅く、解離もかなり遅くなっています。
これにより、kd / kaの比率が同じになり、したがって親和性(KD)も同じになります。
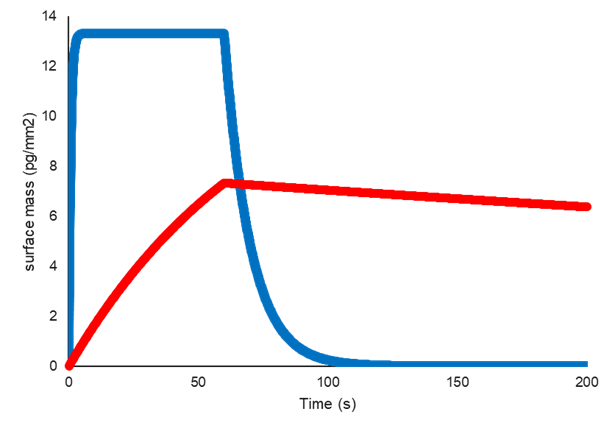
同じ親和性(KD)を持つが反応速度(kaとkd)が異なる2つの相互作用の例
これは、結合親和性の評価だけでは、どの化合物がさらに医薬品として開発する価値があるかを判断するのに十分ではないことを意味します。 研究者は、青線で表される相互作用のように、薬物と標的の間で飽和するまでの迅速な相互作用を探している可能性があります。
薬理学、特に薬物探索において、解離速度は薬物の潜在的な効果を決定する重要なパラメータです。薬物が複合体(すなわち、薬物と薬物標的)にどのくらい滞在するかは、薬物が標的に及ぼす影響、すなわち薬物の効果に影響します。そのため、初期の薬物探索では、潜在的な薬物がその解離速度に基づいてスクリーニングされ、ランク付けされることがよくあります。解離速度(kd、またはkoff)は、バイオセンサーで測定することができます。
WAVEsystemは、結合速度を測定しながら解離定数を測定できる光学バイオセンサーです。WAVEdeltaは、その高感度、粗精製サンプルとの適合性、およびより高いスループットを可能にするwaveRAPID®法により、オフレートスクリーニングに非常に適しています。