The nasal route is commonly used for the delivery of drugs to alleviate a number of local disorders such as sinus congestion and allergic rhinitis. It is also being utilized as an alternative to oral or injection methods for the systemic delivery of a variety of drugs including vaccines, proteins and peptides. Here, the large surface area for absorption in the nose, coupled with the high density of blood vessels, enables efficient absorption into the blood stream. Nasal delivery additionally provides a method for achieving more rapid onset of action compared to oral delivery, as well as avoiding enzyme degradation of the drug within the GI tract and first pass hepatic metabolism. The ease of use for patients of this drug delivery system may also help improve compliance in comparison with other methods available.
A typical nasal spray formulation consists of the drug suspended or dissolved in an aqueous medium, which is filled into a bottle with a metered spray pump. Pump actuation by the patient delivers drug-laden droplets into the nasal cavity. The pump is an integral part of the whole assembly and plays a crucial role in atomizing and delivering an accurate drug dose [1,2]. The properties of the emitted spray plume are thus considered important in assessing the performance of a given product.
This application note introduces the use of the Malvern Spraytec in characterizing the output of nasal pump sprays and discusses the importance of ensuring that the conditions applied during testing mimic patient use as closely as possible.

|
The US Food and Drug Administration's (FDAs), Centre for Drug Evaluation and Research (CDER) has published a draft "Guidance for Industry" document entitled Bioavailability (BA) and Bioequivalence (BE) Studies for Nasal Aerosols and Nasal Sprays for Local Action [3]. This document outlines the testing which may be carried out in order to assess drug bioavailability / bioequivalence during nasal product quality studies in support of new drug applications (NDAs) or abbreviated new drug applications (ANDAs). Within the guidance there is a reliance on the use of comparative in vitro tests in order to determine bioequivalence. These tests may include measurements of the characteristics of the spray plume produced by a given device and formulation, as well as the variation in the delivered dose over the lifetime of the device.
One important consideration for nasal drug delivery is the droplet size produced by the spray pump. Most nasal spray pumps produce droplets in the range from 20µm to around 120µm [4]. The droplet size should be such that the droplets deposit within the nasal passages. If the droplet size is too fine (< 10µm) there is a possibility that the particles will pass through the nasal passages and deposit in the lungs [4]. This could lead to the deposition of drug particles and excipients that are not approved for pulmonary absorption. On the other hand, if the droplet size is too large, the spray may be trapped in the nostrils. It is therefore wise to ensure that the pump produces a spray that has a droplet size within the range stated above during actuation by the patient.
The FDA's draft guidance recommends the use of laser diffraction to determine the droplet size produced for a given device and formulation. Laser diffraction systems, such as the Malvern Spraytec, measure the size of droplets by measuring the intensity of light scattered by particles as a function of angle. Key to the selection of the technique for the characterization of nasal sprays is the ability to acquire this data very rapidly (one measurement can be made every 100µs). This allows the changes in droplet size during pump actuation to be followed in real-time, enabling the atomization dynamics to be assessed.
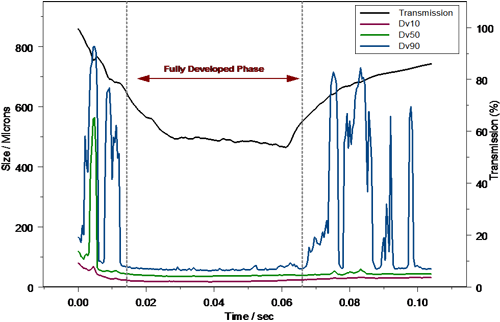
Figure 2 shows a typical measurement of a nasal pump spray produced using the Malvern Spraytec. The rapid data acquisition speed possible using the system allows each phase of spray formation to be detected. At the start of actuation the Formation Phase is observed where the pump produces large particles. This is due to the fact that the liquid flow rate through the spray nozzle is initially quite low. Beyond this the particle size reduces until the Fully Developed Phase is achieved, where the flow rate through the spray nozzle is optimal and the spray droplet size is most stable. Finally, towards the end of actuation, the Dissipation Phase is observed where, due to the emptying of the metering chamber within the device, the flow rate decreases causing the formation of large droplets. Complete actuation of a nasal spray is rapid and occurred in less than 130ms in this case.
|
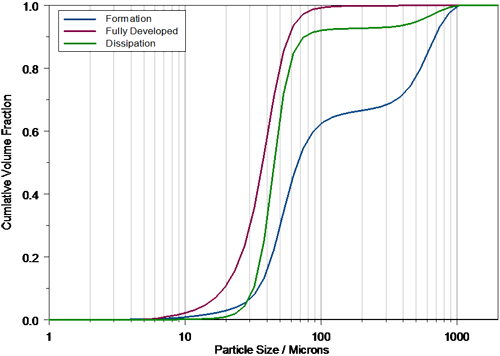
FDA guidelines recommend that, in order to characterize nasal spray delivery, the data collected during the fully developed phase should be analyzed. Definition of the fully developed phase allows for a statistically valid comparison between Test and Reference List Drug (RLD) products and is therefore an important part of data analysis. In the case of the example above, the formation phase is obvious within the profile, and can be defined within the Spraytec software on the basis of a time window during which the reported Dv10, Dv50 and Dv90 is stable. Based on this time window, the average particle size for each phase can be calculated (figure 3), and the reproducibility of delivery during the fully developed phase assessed.
|
Accurate spray characterization is obviously important for effective nasal spray testing, but equally critical is the need for representative spray pump actuation [5]. During nasal spray pump testing using the Spraytec system, the variance associated with manual actuation pump can be eliminated by using automated actuation stations. These typically control either the force or velocity of actuation, ensuring that a specific, reproducible actuation profile is followed during all of the tests outlined by the FDA. Indeed, the FDA recommends that automated actuators be used in order to ensure that operator bias is eliminated, allowing variations between different pumps and formulations to be readily observed. However, care must be taken to ensure that the chosen actuation profile is representative of the manual actuation profile of the target patient group if the in vitro tests are to be a good mimic of in vivo performance. This requires some understanding of how the pump performs under different actuation conditions.
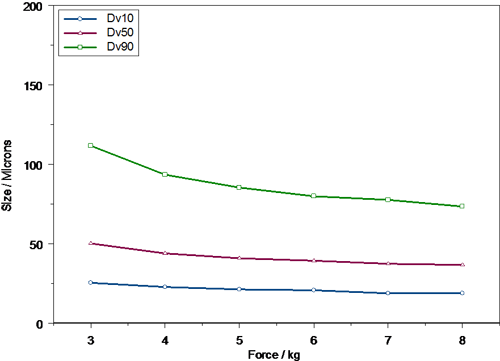
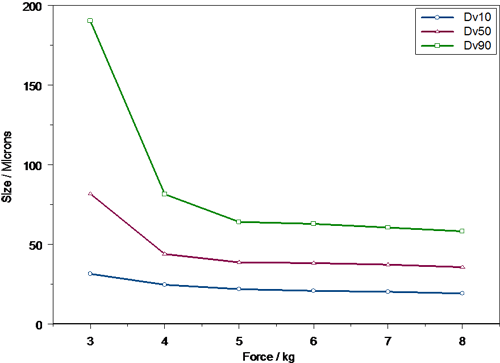
An example of how automated actuation stations can be used to study the effect of actuation variance on droplet size is shown in figures 4 and 5. Here the Dv10, Dv50 and Dv90 are shown for the same formulation delivered using two commercially available pump systems. In this case the force of actuation was varied and the droplet size delivered during the fully developed phase of the spray event recorded. The results indicate clearly that spray droplet size is a function of actuation force and also that this dependence is pump specific. With low actuation forces both pumps produce relatively large, broad droplet size distributions. However, the particle size observed for Pump B (figure 5) is much coarser than for Pump A (figure 4), with a significant amount of material being present above 100 μm in size. This would suggest that Pump A may not be ideal for those patients who cannot actuate a pump strongly, for instance older patients.
|
|
As actuation force is increased, the droplet size decreases and the droplet size distribution width, as assessed from the difference between the Dv10 and Dv90, becomes narrower. For both pumps the dependency of droplet size on actuation force decreases markedly above 5 kg, but pump B is noticeably less sensitive to actuation force beyond this point. This lack of sensitivity may be advantageous, as this pump will deliver a similar particle size irrespective of the patent if a large actuation force can be achieved.
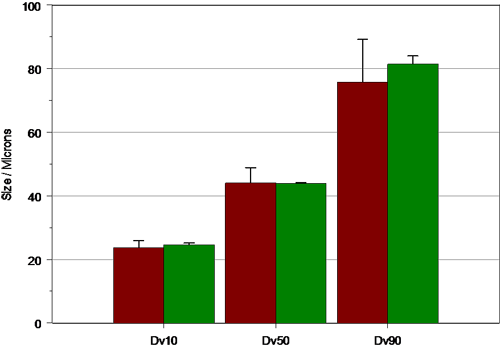
Once the behavior of the pump has been understood, the actuation parameters that best mimic patient use can be selected. This is shown in figure 6, where the results obtained for manual actuation of pump B by an elderly patient are compared to those obtained using automated actuation at a force of 3 kg. As can be seen, the particle size observed in each case is comparable. However, the variance observed during the measurements made using the automated actuation station is significantly smaller.
|
Nasal pump sprays and aerosols are becoming increasingly popular as drug delivery systems because of the advantages associated with drug absorption within the nose. This is reflected in the publication of the FDA's guidance document, which has set out the in vitro testing required in order to assess whether consistent drug delivery can be achieved for a given product. Techniques such as laser diffraction provide an extremely powerful tool for understanding how drug delivery is achieved. However, it is important that the target patient group is kept in mind during product testing if realistic results are to be obtained. This requires the use of the correct pump actuation conditions.