Aggregation of both biological and synthetic materials is a source of serious concern, posing problems at various stages of the discovery and formulation processes. Liposomes have been used in drug discovery and drug delivery for some time, and the biophysical characterization of these systems and their payloads is critical to understanding their function. One such payload is the membrane-associated receptor tyrosine kinase (RTK) targets which continue to be the focus of many discovery campaigns. RTKs are typically single-pass transmembrane signaling proteins which are difficult to purify intact, and are important targets in many disease pathways. Our aim is to correlate previously published activity data with the changes in size and aggregation state of the liposome-RTK complex as measured by Nanoparticle Tracking Analysis (NTA) by the NanoSight product range from Malvern Instruments.
Aggregation of both biological and synthetic materials is a source of serious concern, posing problems at various stages of the discovery and formulation processes. The ability to identify and characterize aggregates is of broad utility, and the experiments described here demonstrate the unique ability of the Nanoparticle Tracking Analysis (NTA) technique to understand the dynamics of aggregation in a biological system through high resolution particle-by-particle analysis.
For the work presented in this whitepaper, a model system was utilized that has been used in drug discovery research and presents some unique challenges in reagent characterization. This system was generated to allow functional assembly of cloned/recombinant membrane receptors on liposomes, without the necessity of reconstitution of full length receptors. The aim was to correlate previously published activity data1,2 with the changes in size and aggregation state of the liposome-RTK complex measured by Nanoparticle Tracking Analysis (NTA). It is expected that the experiments described here represent characterization methods of broad utility to the nanoparticle field. Determination of size, assessment of polydispersity, and identification of components which promote aggregation are all of interest and can all be assessed via these simple experiments.
Liposomes have been used in drug discovery and drug delivery for some time, and the biophysical characterization of these systems and their payloads is critical to understanding their function. One such payload, which we have investigated here, is the membrane-associated receptor tyrosine kinase (RTK) targets which continue to be the focus of many discovery campaigns. RTKs are typically single-pass transmembrane signaling proteins which are difficult to purify intact, and are important targets in many disease pathways. This model characterization system was chosen here because of the abundance of activity data that exists.
For this work, we first investigated the aggregation state of a representative RTK target (kinase domain of insulin receptor). We then analyzed the RTK on an engineered cell-like scaffold, specifically a liposome functionalized with nickel-chelated head groups (described in references 1 and 2). Briefly, the nickel head groups facilitate template-directed self-assembly of His-tagged RTKs, such that the kinase domains (cytoplasmic fragments) are oriented as though within the cell (Figure 1). This analysis was performed using magnesium (Mg) or manganese (Mn) as the available counter-ion in the assay buffer, in order to understand why manganese might cause an increase in the activity level of kinases, despite the understanding that manganese is not a physiologically-relevant counter-ion. We hoped to gain insight into the effects of counter-ions using NTA, and to correlate this to previously obtained activity data.
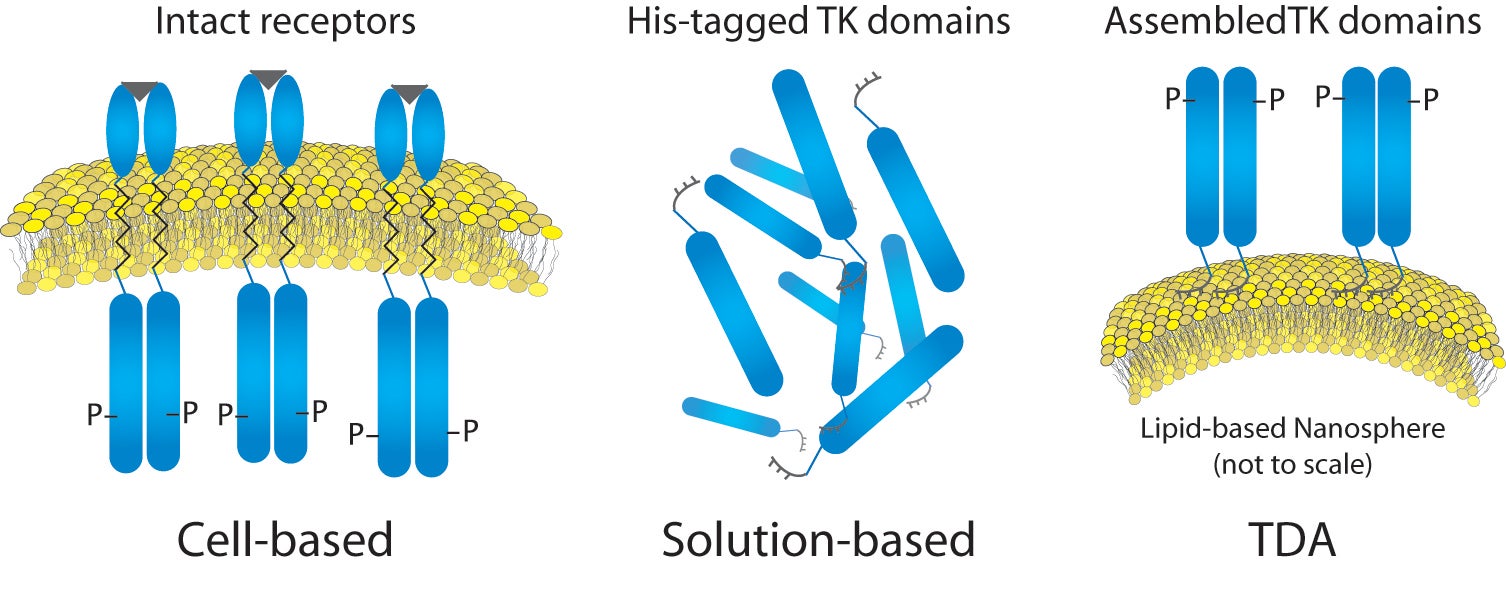
Figure 1: Liposome- Insulin Receptor Complexes
The NanoSight range of instruments from Malvern Instruments offers a unique capability to rapidly characterize this well-studied drug discovery system by directly visualizing nanoscale particles in suspension (between 10 nm and 2000 nm) with high-resolution, in real-time and with minimal sample preparation. The Nanoparticle Tracking Analysis (NTA) technique delivers an unprecedented insight into size distributions, using a particle-by-particle approach to particle sizing. NanoSight provides visual validation and high resolution analysis of particles within the nanoscale range with both size and concentration measurements. NTA offers a robust analysis of this liposome-protein system and sheds light on this method of kinase activation.
Extruded liposomes and purified protein were obtained from Blue Sky BioServices (Worcester, MA, USA). Samples were diluted in HEPES buffer prior to analysis by a NanoSight NS300 equipped with a high sensitivity Hamamatsu sCMOS camera, 20x objective lens, and a 50 mW green 532 nm laser. Samples were analyzed using NTA 3.1 Build 46 software. NanoSight technology observes the Brownian motion of particles in suspension and calculates their size through the Stokes-Einstein equation. It derives concentration by observing particles on a frame-by-frame basis through the use of the sCMOS camera (25 frames per second). The software determines the average number of particles per frame in the field of view and calculates a number-weighted concentration result.
Nanoparticle Tracking Analysis afforded an appreciation of the rapid kinetics with high resolution analysis of aggregation on a real-time basis. We could see a simultaneous decrease in concentration and an increase in size over time in the presence of manganese buffer. The liposomes clearly aggregate in the presence of Mn (Figures 2 and 4). Analysis of the insulin receptor RTK domain in HEPES (or Tris) buffer suggests that they further aggregate in presence of manganese (Figure 3 and 4) This analysis was challenging because the protein itself appears aggregated to a degree in the absence of either counter-ion when noting the initial time curve in both Figures 3 and 4, although there is clearly a significant increase in aggregate size in the presence of manganese that is not observed with addition of magnesium.
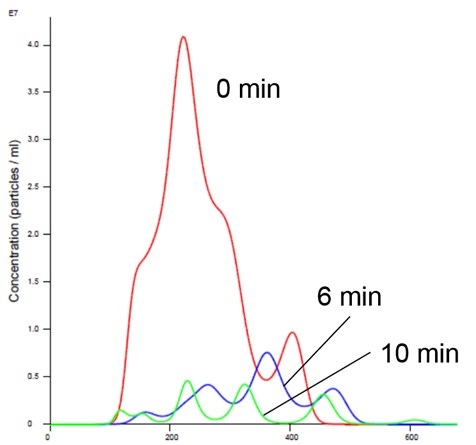
Figure 2: Liposomes in Mn Buffer show aggregation (increased size and decreased concentration)
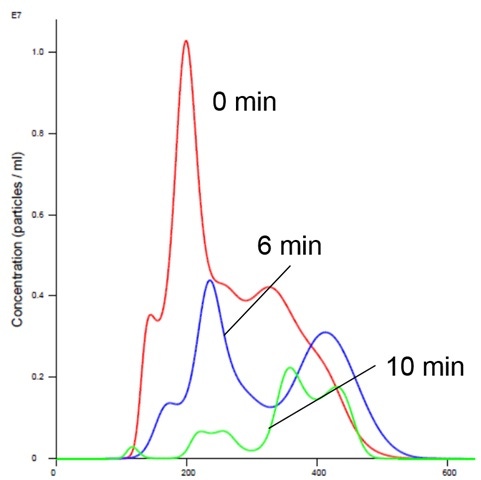
Figure 3: Insulin receptor alone in Mn buffer showing aggregation (increased size and decreased concentration)
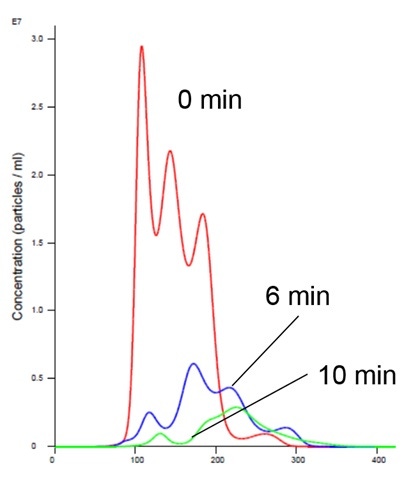
Figure 4: Insulin receptor and liposomes in Mn buffer showing aggregation (increased size and decreased concentration)
Addition of magnesium to RTK does not cause aggregation and is consistent with previously-generated data which shows no appreciable activity in the presence of Mg1,2,5 (Figures 5 and 6).
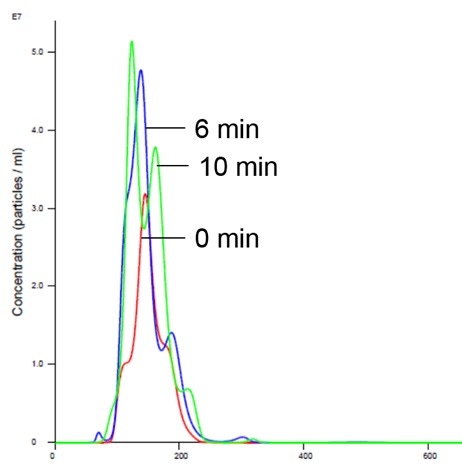
Figure 5: Liposomes in Mg buffer show stabilized size and concentration
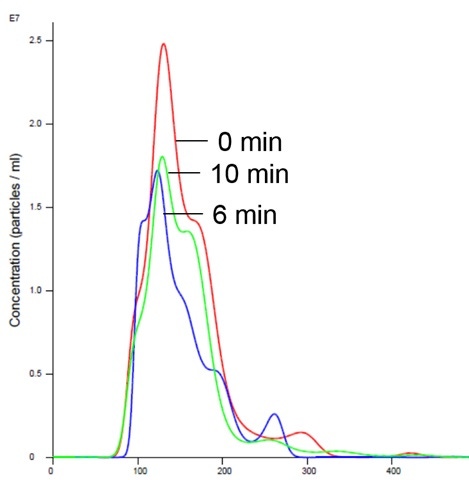
Figure 6: Insulin receptor and liposomes in Mg buffer show stabilized size and concentration
The hypothesis that magnesium is a biologically relevant counter-ion but does not lead to activity is puzzling. Adding Mg to RTK and liposomes did not promote aggregation but does lead to high cell like activity1,2,5. It is thought that the RTK assembles on the outside of the liposome in the same manner as inside the cell (Figure 1).
Addition of Mn causes aggregation and increases activity in a cost-effective and simple manner, with reasonable reproducibility and robustness. When RTK is properly self-assembled on a liposome in the presence of magnesium, a biologically relevant counter-ion, the following benefits are reported1,2,5:
a. increase in autophosphorylation and substrate phosphorylation
b. highest catalytic rates (except where autophosphorylation is self-inhibitory, Tie2)
c. improved substrate selectivity
Template-assembled enzymes are expected to be a better screening tool because the target appears to be in cell-like conformation, allowing a unique opportunity for screening protein-protein interactions. This study points to the need for better understanding of the difference between forced colocalization (or molecular crowding) and functional assembly of RTK complexes (for interesting colocalization discussion, see reference 4). The work presented here is consistent with the hypothesis that manganese increases the catalytic activity of some kinases by facilitating formation of aggregated complexes.
NTA is a useful technique for accurately characterizing the aggregation behavior of liposome preparations under a range of conditions. The high resolution data provides a unique insight into the aggregation kinetics of these systems. The results here call into question the relevance of compounds selected in high-throughput screening (HTS) assays using Mn as a counter-ion.
(1) Esposito EA, Shrout AL, Weis RM. J Biomol Screen 2008; 13 (8): 810-6.
(2) Shrout AL, Esposito EA, Weirs RM. Chem Biol Drug Des 2008; 71 (3): 278–281.
(3) Langmuir, 2007. 23(6): p. 3280-9.
(4) Curr Opin Struct Biol, 2006. 16(6): p. 702-9.
(5) Gridley S, Shrout AL, Esposito EA, Prog. Mol. Bio. Transl. Sci. 2010; 91:209-39
TDA technology used to prepare the liposomes is protected by patent and available via Blue Sky Bioservices for license: US: 8,541,190.