


The PEAQ-ITC, a MicroCal technology, is a highly sensitive, low volume isothermal titration calorimeter for the label-free in solution study of biomolecular interactions. It delivers direct measurement of all binding parameters in a single experiment and can analyze weak to high affinity binders, using as little as 10µg sample. Semi-automated maintenance minimizes operator intervention and the system is upgradable to the fully automated PEAQ-ITC Automated, making it ideal for laboratories where speed, sensitivity and the ability to accommodate higher workloads in the future are paramount.
Select a model:
Looking for more information?
To request a quote, more information or download a brochure select an option below.
MicroCal PEAQ-ITC is designed for ease of use and exceptional sensitivity. The system directly measures the heat released or absorbed during biochemical binding events, from which it calculates binding affinity (KD), stoichiometry (n), enthalpy (ΔH), and entropy (ΔS).
With minimal sample preparation and system optimization data is generated quickly and easily. The wide affinity range enables analysis of weak to high affinity binders, with excellent reproducibility.
MicroCal ITC is an essential tool for any research laboratory studying biomolecular interactions where high sensitivity and fast results are paramount.
A wide range of applications can be investigated with MicroCal PEAQ-ITC, including the characterization of molecular interactions of small molecules, proteins, antibodies, nucleic acids, lipids and other biomolecules. It can also be used to measure enzyme kinetics.
MicroCal PEAQ-ITC analysis software offers experiment design simulation, batch evaluation of large data sets, automated assessment of data quality and a streamlined user interface that guides the user to final figures and presentation quality graphs quickly and easily.
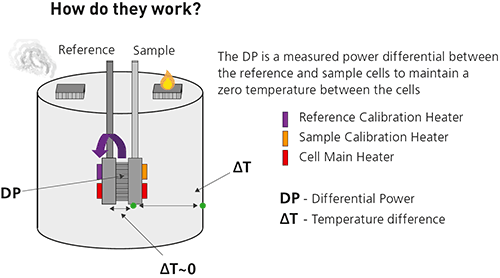
Isothermal titration microcalorimeters measure the heat change that occurs when two molecules interact.
Heat is released or absorbed as a result of the redistribution and formation of non-covalent bonds when the interacting molecules go from the free to the bound state. ITC monitors these heat changes by measuring the differential power, applied to the cell heaters, required to maintain zero temperature difference between the reference and sample cells as the binding partners are mixed.
The reference cell usually contains water, while the sample cell contains one of the binding partners (the sample, often but not necessarily a macromolecule) and a stirring syringe which holds the other binding partner (the ligand). The ligand is injected into the sample cell, typically in 0.5 to 2 μL aliquots, until the ligand concentration is two- to three-fold greater than the sample.
Each injection of ligand results in a heat pulse that is integrated with respect to time and normalized for concentration to generate a titration curve of kcal/mol vs molar ratio (ligand/sample). The resulting isotherm is fitted to a binding model to generate the affinity (KD), stoichiometry (n) and enthalpy of interaction (ΔH).
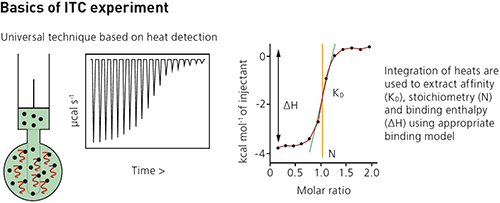
| Measurement type | Affinity (KD)
Enthalpy ∆H Entropy ∆S Stoichiometry (n) |
|---|---|
| Sample volume | 280 µL |
| Cell volume | 200 µL |
| Injection syringe volume | 40 µL |
| Injection volume precision | < 1% @ 2 µL |
| Sample throughput | 0-12 per 8 h day |
| Cell material | Hastelloy |
| Cell | Coin-shaped |
| Noise | 0.15 ncal/s |
| Temperature range | 2 °C to 80 °C |
| Temperature stability | ± 0.00012°C |
| Response time | 8 s* |
| Multiple feedback modes | Yes (passive, high gain, low gain) |
| Notes | *The PEAQ-ITC Instrument Response Time is a true time constant. It is the time interval between the first deviation away from the baseline, and the point on the peak that is 63% of the maximum peak height. |
| Operating temperature (°C) | 10 °C to 28 °C |
|---|---|
| Humidity | 0% to 70% RH, non condensing |
| Power | 100 - 240 VAC (power adapter), 24 VDC (power supply to instrument)
50/60 Hz, 150 W |
|---|---|
| Weight | 13.6 kg |
| Dimensions (W, D, H) | 43 x 38 x 46 cm (calorimeter + wash station) |
MicroCal PEAQ-ITC instruments provide value to a wide range of applications, some of which are outlined below.
ITC is ideal for measuring very accurate affinities and stoichiometries of biological interactions. It does not require the labeling or immobilization of the interacting molecules and has no molecular mass limitations. There are thousands of articles that have used ITC to study biological interactions involving proteins, nucleic acids, antibodies, carbohydrates, lipids ATP, inhibitors and drug compounds to name some of the more common examples.
Where does the ligand bind the target? At the active site? At the allosteric site? Is the interaction competitive, non-competitive or uncompetitive with respect to other ligands and cofactors? Is the binding cooperative?
ITC can give you an insight into these mechanisms of binding and interaction.
Do you have a simple assay for your enzyme? Does your assay involve a complex assay set up? Does modifying your substrate interfere with your SAR or mechanistic studies?
ITC can be used to study any enzyme reaction and requires only nano to picogrammes of material AND is label-free.
ITC has been used extensively for the determination of CMCs for a great variety of micellar complexes including surfactants, block copolymers, surface-active peptides and proteins, as well as of amino-acid-type surfactants and amino-functionalized lipids.

| Title | Version | Date | Download |
|---|---|---|---|
| {{ row.title }} | {{ row.softwareVersion }} | {{ row.date }} | Download |
Sorry, no manuals are available for this product
| Title | Version | Date | Download |
|---|---|---|---|
| {{ row.title }} | {{ row.softwareVersion }} | {{ row.date }} | Download |
Sorry, no software downloads are available for this product
|
|
Unmatched uptime and performance
Malvern Panalytical instruments are built to last and good maintenance and care ensure continued peak performance for many years to come. Service Agreements build this into your schedule and budget, based on preventive maintenance and remote support . Different levels include repair visits, response times and parts coverage as needed. OQ services, training and application consultancy can be added as well.
|
|
|
|
Advanced analysis and actionable insights
|
|
|
|
Easy start and streamlined routine operations
|
|
|
|
Want to speak to a service expert or find the services that are just right for you?
Reach out to our experts and evaluate your services needs and business risks by answering a few questions around the usage of your Malvern Panalytical instrument.
|
|
|
*Please note that the exact details of our services and their availability are determined by a variety of customer-specific factors, such as product type, product configuration, or location. |
||

Direct, label free and in-solution measurements of binding affinity and thermodynamic parameters in a single experiment to meet the varied and demanding requirements of today’s life science laboratories.