The increased demand for portable electronic devices, including mobile phones, laptops and new ‘wearables’, has required advances in battery technology to provide a low-cost, lightweight, long-lasting and stable power source. With fossil fuels dwindling and CO2 regulations becoming more stringent, battery technology is increasingly being used in applications such as renewable energy storage and electric vehicles, which require ever more lightweight, safe, high-power and fast-charging batteries.
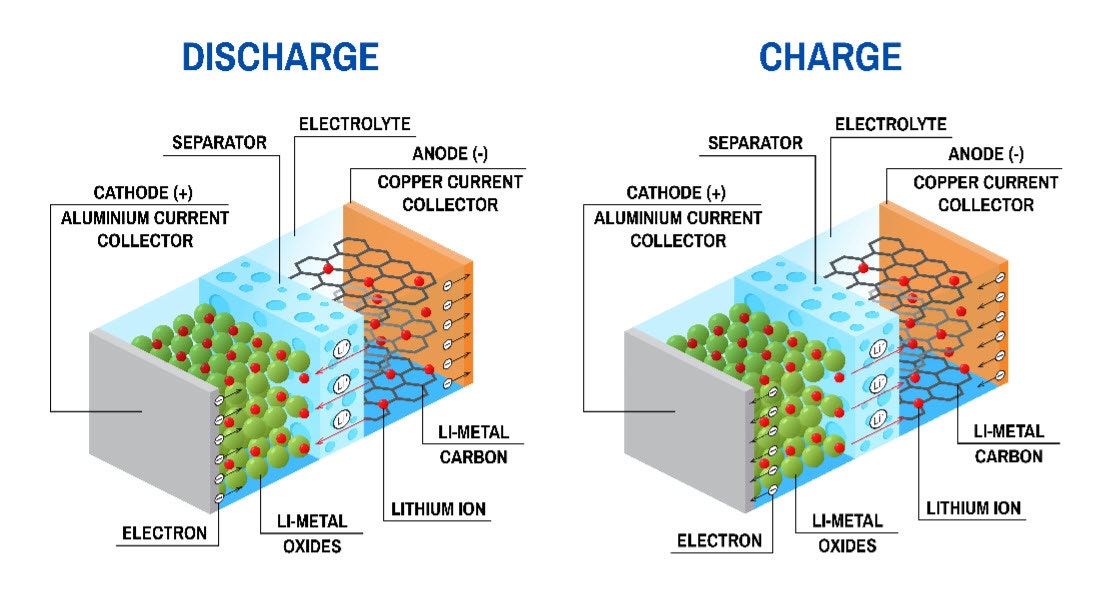
Schematic of a Li-ion battery. During discharge, lithium ions (Li+) move from the anode to the cathode, conducted via the electrolyte, with flow in the reverse direction occurring when the battery is charged. Anodes are typically graphite-based, while cathodes are often manufactured using lithium iron phosphate, lithium cobalt oxide, lithium-manganese oxide, lithium-nickel cobalt manganese oxide, etc.
The cornerstones of battery performance are power, which impacts current and discharge characteristics, and energy storage capacity. Battery power is determined by the rate of reaction between the electrodes and the electrolyte, while storage capacity is a function of the volume of electrolyte within the cell. These properties are intrinsically linked to the intercalation structure and primary particle size of the electrode particles, which determine how well the mobile ions are taken up and released by the electrode1. Particle size distribution and particle shape also influence particle packing and hence the volume of electrolyte that can be accommodated within the interstitial voids of the electrode, which affects storage capacity.
Login or create an account to read more about this case study.
The increased demand for portable electronic devices, including mobile phones, laptops and new ‘wearables’, has required advances in battery technology to provide a low-cost, lightweight, long-lasting and stable power source. With fossil fuels dwindling and CO2 regulations becoming more stringent, battery technology is increasingly being used in applications such as renewable energy storage and electric vehicles, which require ever more lightweight, safe, high-power and fast-charging batteries.

Schematic of a Li-ion battery. During discharge, lithium ions (Li+) move from the anode to the cathode, conducted via the electrolyte, with flow in the reverse direction occurring when the battery is charged. Anodes are typically graphite-based, while cathodes are often manufactured using lithium iron phosphate, lithium cobalt oxide, lithium-manganese oxide, lithium-nickel cobalt manganese oxide, etc.
The cornerstones of battery performance are power, which impacts current and discharge characteristics, and energy storage capacity. Battery power is determined by the rate of reaction between the electrodes and the electrolyte, while storage capacity is a function of the volume of electrolyte within the cell. These properties are intrinsically linked to the intercalation structure and primary particle size of the electrode particles, which determine how well the mobile ions are taken up and released by the electrode1. Particle size distribution and particle shape also influence particle packing and hence the volume of electrolyte that can be accommodated within the interstitial voids of the electrode, which affects storage capacity.
Particle sizing of electrode materials is commonly performed using laser diffraction technology, such as the Mastersizer 3000; although the Zetasizer Pro and Zetasizer Ultra can also be employed for analyzing the much smaller particles used for electrode materials or separators. Automated imaging (Morphologi 4) is commonly employed for particle shape analysis but can also be coupled with Raman spectroscopy to give particle-specific structural information. The primary tool for studying the structure of electrode materials, however, is X-ray diffraction (XRD) with Aeris or Empyrean diffractometers widely used in this sector. Empyrean also allows small-angle X-ray scattering (SAXS), an important tool for evaluating the primary particle size or crystallite size of nanoparticle electrodes which tend to exist in aggregated form.
Manufacture of the cathodes and anodes involves mixing the active electrode material with some form of conductive additive such as carbon (e.g. carbon black and/or graphite) and a polymeric binder dissolved in a solvent to form a slurry. The slurry/suspension is then applied to a metal foil in a continuous coating process and the solvent driven off to produce a dried coating which is subjected to calendaring, a compression process that involves feeding the coated foil through a series of rollers. The electrodes are then punched or cut to size prior to winding or stacking between the separator film before inserting into the case, wetting with electrolyte, and sealing. The cells are then activated over several weeks through a series of charge/ discharge cycles.
These processes exert a significant influence on electrode structure and are related to the rheological, or flow properties, of the battery slurry. These properties can be influenced by the extent to which raw materials are dispersed during the slurry manufacturing process, affecting the size of particles deposited on the foil or the impact of compressive forces applied during calendaring, which affects the porosity of the finished coating. These are not discrete effects - rather changes made during one step can have an impact on consecutive processes, as well as the properties of the finished electrode2.
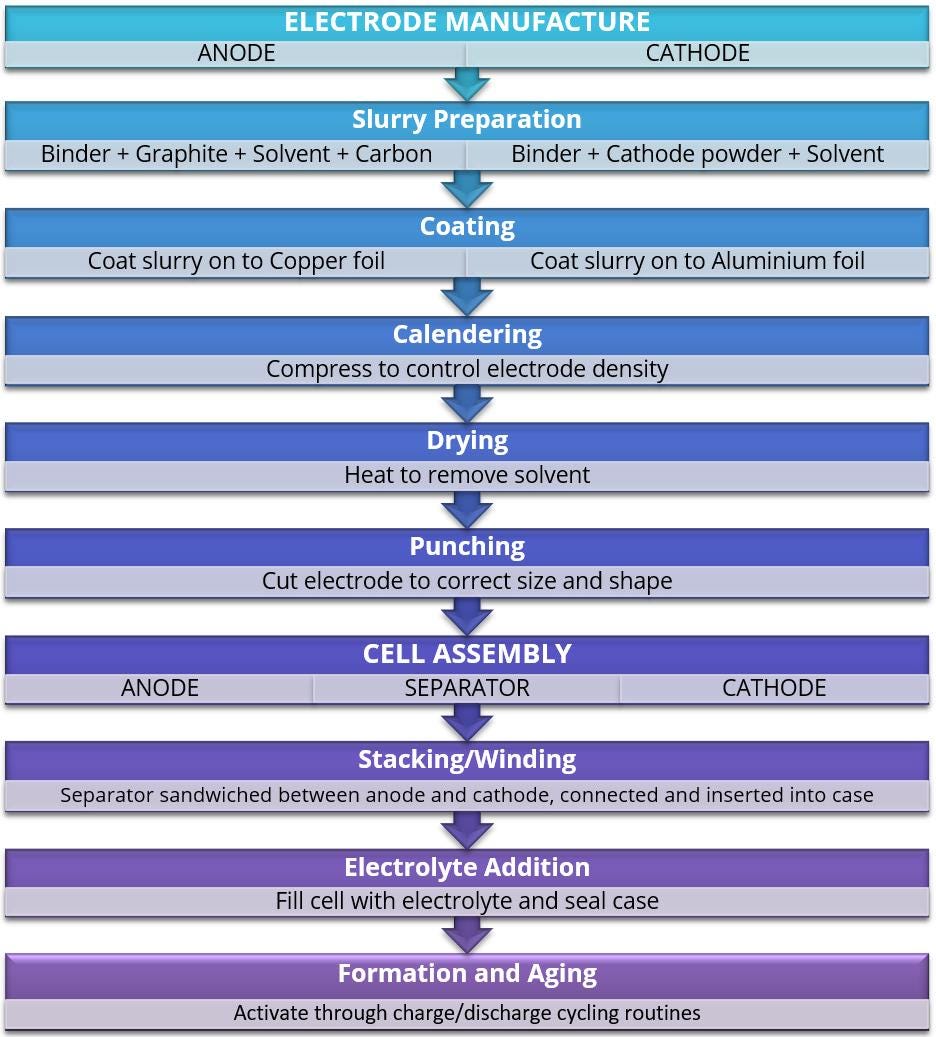
The manufacture of Li-ion battery electrodes is a multi-step process with each step having potential to influence the electrochemical performance of the finished device2
Slurry rheology is influenced by the size, shape, concentration and charge of dispersed components, hence it is important to optimize electrode particle properties not just for their individual electrochemical performance but for the electrode manufacturing process as a whole3. Ultimately it is the performance, and safety, of the final battery cell which is of critical importance. The most common tool for measuring the general rheology of battery slurries is a rotational rheometer, such as Kinexus, although a capillary rheometer such as the Rosand RH7 or RH10 may better represent the electrode coating process4.
So, what happens to the electrode structure during initial formation, as well as the repeated charge/discharge cycling of the fabricated battery cell? The repeated transfer of charge-carrying ions in and out of the electrode lattice puts strain on the cell structure and can induce unwanted phase changes, even more so if fast charging and/or discharging is required. Deterioration in performance is most commonly due to the build-up of defects within the electrode which hinders ion mobility. The mechanism of these processes on the electrode structure can be studied with in-operando XRD, using hard X-rays (Mo or Ag Kα) capable of penetrating a battery cell, ideally (but not limited to) a pouch cell5.
It is then possible to correlate variations in the crystallographic structure of the electrodes with the amount of Li incorporated into them. The spatial resolution of in-operando transmission XRD (approx. 1 mm) also makes it an ideal tool for probing localized wear, since cells close to the connector tend to degrade faster than those further away. It can also identify areas within the cell that are not functioning properly due to aging or incomplete wetting with electrolyte. Empyrean equipped with the GaliPIX3D detector is ideally suited for this application since it offers the highest resolution data in the shortest possible time frame.
References
1) Julien, C.M., Mauger, A., Vijh, A., Zaghib, K.; Lithium Batteries: Science and Technology; Springer: Heidelberg, Germany, 2015
2) Bockholt, H. et al. ‘The interaction of consecutive process steps in the manufacture of lithium-ion battery electrodes with regard to structural and electrochemical properties’; Journal of Power Sources 325 (2016) 140-151
3) White paper: Exploring the impact of particle characteristics on suspension rheology
4) White paper: Establishing an analytical toolkit for the optimization of Li-ion battery electrode manufacturing
5) Application note: High-quality in-operando X-ray diffraction analysis of pouch bag lithium-ion batteries